Written by Jesse McLaren
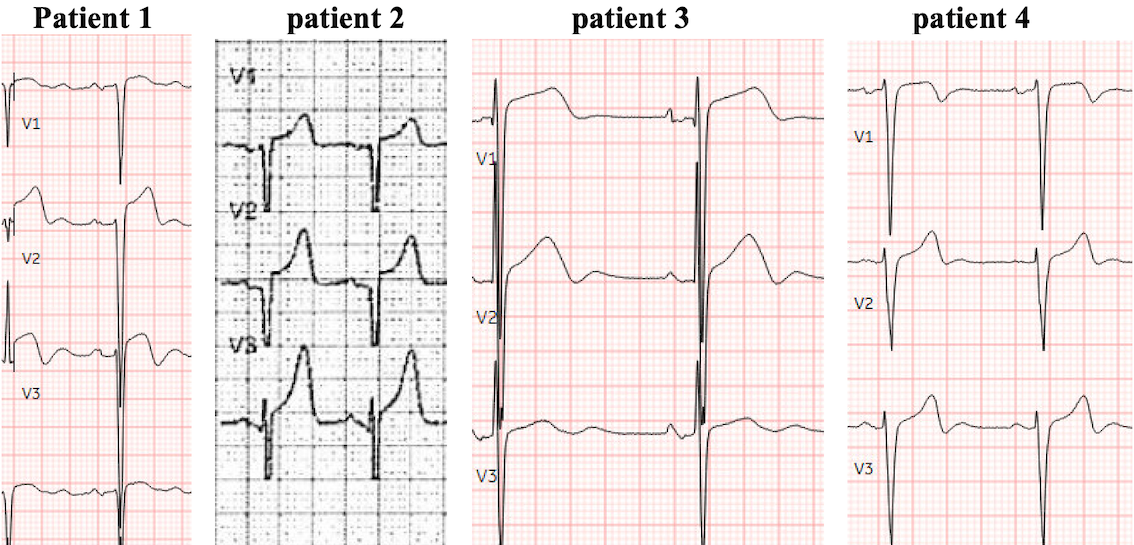
Four patients presented with chest pain, an ECG showing LVH, and an elevated troponin. Which patient has an acute coronary occlusion?
Patient 1: 50 year old, no past medical history, with chest pain that began on exertion but continues, and feels like heartburn
–
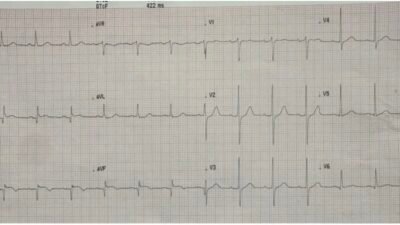
Patient 2: Prehospital ECG on a 55 year old with no past medical history, with exertional chest pain and syncope, EMS Code STEMI
–
Patient 3: 60 year old, history of hypertension, with one week of chest pain and shortness of breath on exertion
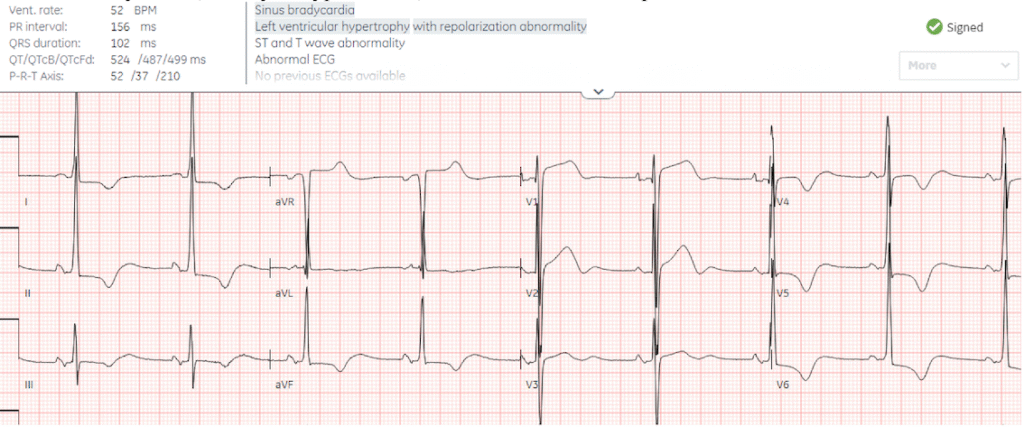
–
Patient 4: 55 year old, no past medical history, with 3 days of exertional chest pain
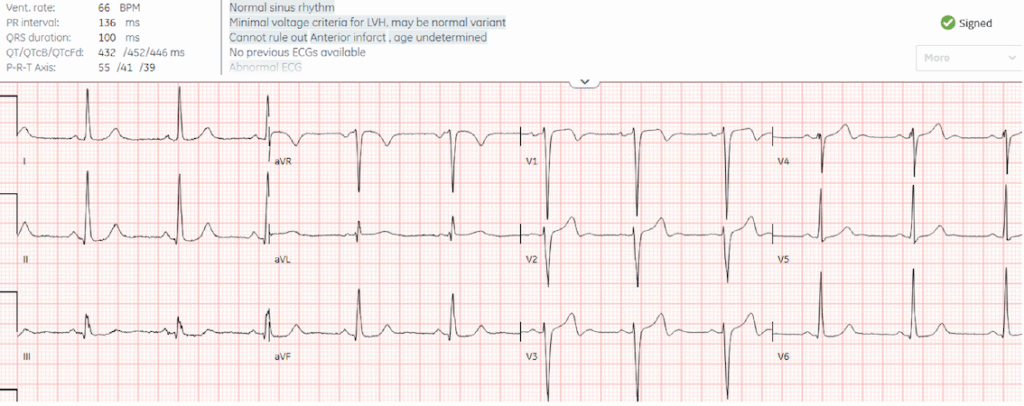
–
LVH: STEMI vs OMI paradigms
LVH causes secondary ST/T changes that are discordant and proportional to the QRS, which can mimic STEMI and Wellens syndrome. But because STEMI criteria measure ST segment elevation in isolation (ignoring ECG principles of totality and proportionality), there is no way to differentiate LVH with vs without acute coronary occlusion. So STEMI is defined as ST elevation in the absence of LVH. But this creates an obvious diagnostic dilemma of how to identify acute coronary occlusion in the presence of LVH. This leads to false positive STEMI leading to unnecessary cath lab activation (especially with prehospital ECG machines that truncate voltages), and false negative STEMI with delayed reperfusion. This is compounded by discharge diagnosis of “STEMI” vs “NSTEMI” that reflects time to treatment more than STEMI criteria. (Alencar, Meyers, Frick, McLaren, Smith. Door-to-balloon time outperforms ST-segment elevation in predicting the STEMI vs NSTEMI final diagnosis. J Clin Med 2025)
Armstrong proposed that STE/RS>25% could identify LVH + STEMI, but in leads with large S waves this requires a massive amount of ST elevation and will miss subtle occlusions. (See Dr. Smith’s post which shows the fatal flaws of the Armstrong study). The initial troponin is often not helpful in this dilemma, because it can be falsely reassuring in acute coronary occlusion, or elevated from hypertensive heart disease in the absence of acute coronary occlusion. But using totality and proportionality, the Queen of Hearts has been trained to differentiate LVH with vs without OMI, which can help reduce false positive STEMI activations and identify LVH + OMI. Let’s see how patients were managed, and how the Queen performed
Patient 1: 50 year old, no past medical history, with chest pain that began on exertion but continues, and feels like heartburn

PMcardio for Individuals now includes the latest Queen of Hearts model, AI explainability (blue heatmaps), and %LV Ejection Fraction. Download now for iOS or Android: https://individuals.pmcardio.com/app/promo?code=DRSMITH20. As a member of our community, you can use the code DRSMITH20 to get an exclusive 20% off your first year of the annual subscription. Disclaimer: PMcardio is CE-certified for marketing in the European Union and the United Kingdom. PMcardio technology has not yet been cleared by the US Food and Drug Administration (FDA) for clinical use in the USA.
Back to Patient 1:
There’s LVH with an S wave in V2 of 35, which would require 9mm of ST elevation to meet Armstrong criteria.
Smith: Unlike the Smith criteria for LBBB, the Armstrong “criteria” for LVH were developed with a very poor methodology and are very INsensitive (not sensitive enough). In this case, their 25% rule works for lead V3 (3/11 = 27%), and the rule requires only one lead to be more than 25%.
Queen of Hearts identified LAD OMI with ECG signs of reperfusion, but clinically the patient still had pain at the time of the ECG. The ECG was repeated 15 minutes later, when the patient reported reduction but still ongoing pain
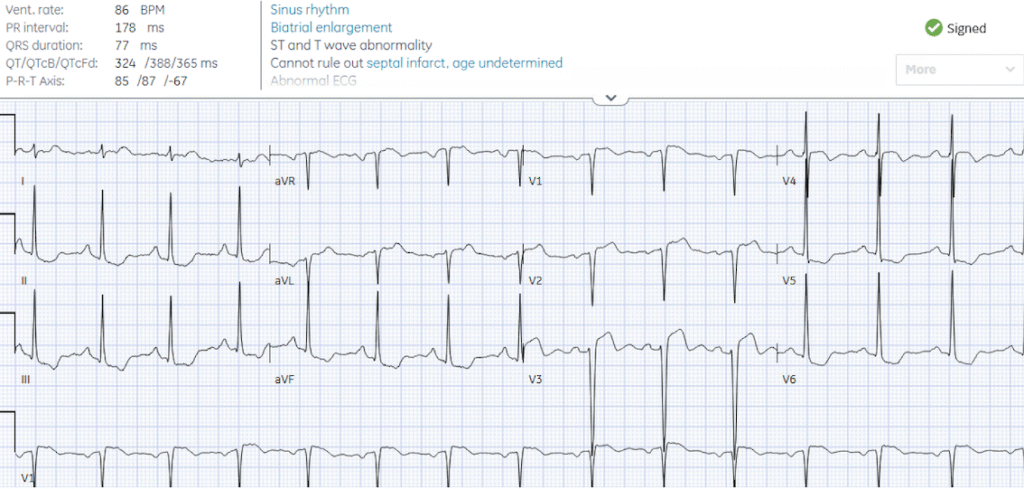
Less STE in V2 but also less reperfusion in V3, and greater inferolateral STD.
Smith: in this ECG, there is no lead that meets the 25% rule. V2 is 2/13 = 15.4% V3 is 4/25 = 16%. My anecdotal rule for LAD OMI in the setting of LVH (deep S-wave in V1-V3-4) is that any STE more than 15% is very suspicious.
Smith continues: I have tried to study this topic, but it is very difficult because it is very hard to find cases of LVH, as manifested by deep S-waves in V1 to V3-4. Such cases are very uncommon. Why are they uncommon? I believe, based on anecdotal evidence, that they are uncommon because when a patient has pre-existing ECG LVH with deep S-waves, LAD occlusion obliterates the S-wave voltage, similar to terminal QRS distortion in patients with normal voltage.
Queen now calls this OMI:
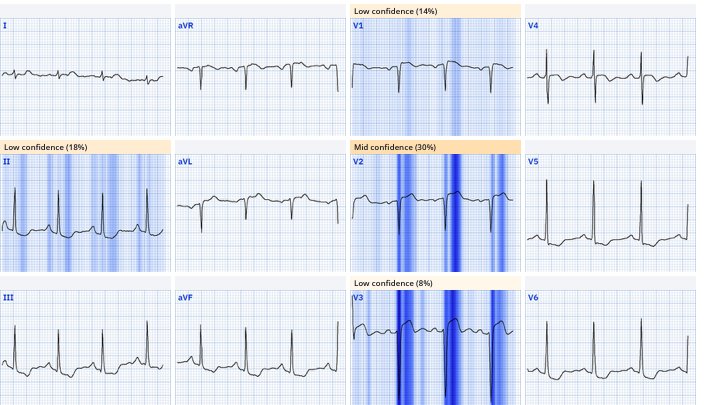
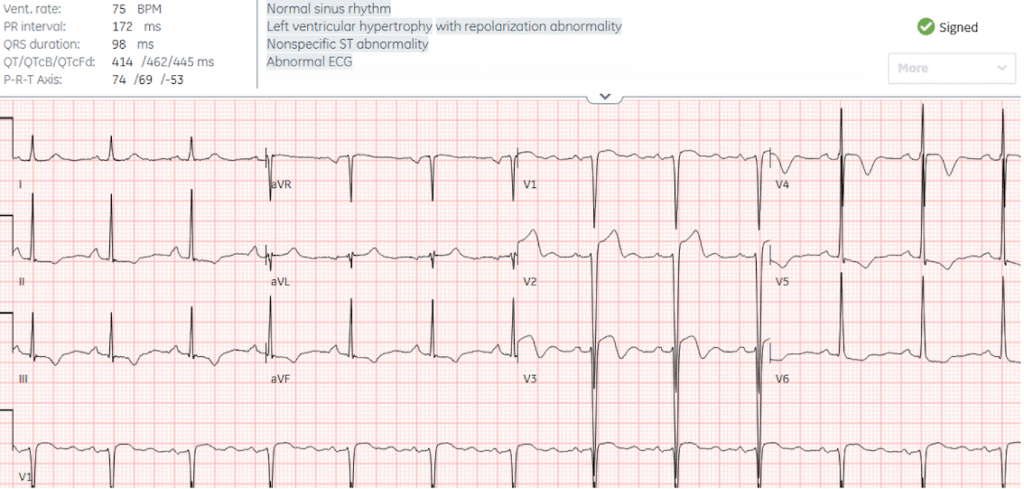
Cath lab was activated: 100% mid LAD occlusion, with RCA CTO and 80% circumflex stenosis. LAD stented. First troponin I was 2,000 ng/L and peak 10,000ng/L. Discharge ECG showed reperfusion T wave inversion:
Discharge diagnosis was “STEMI” because of rapid reperfusion, even though no ECG had a final interpretation of STEMI, because STEMI criteria cannot be applied in the presence of LVH.
Smith: only the most academic of physicians are aware that the “STEMI millimeter criteria” exclude LVH. That is fine with me.
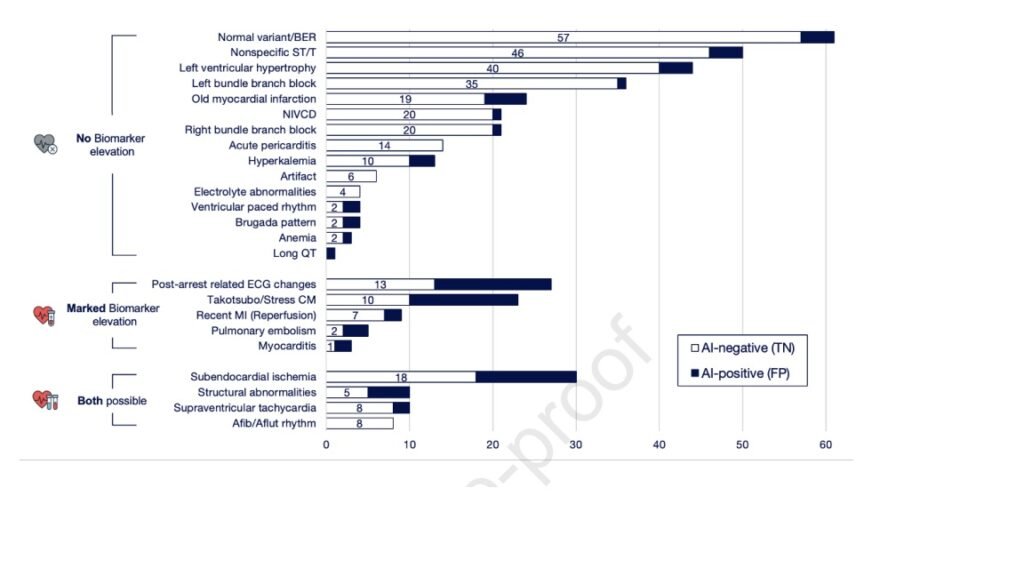
The Queen of Hearts knows when the ST Elevation of LVH is NOT due to OMI. In our recent study from JACC: Cardiovascular Interventions, AI-Enabled ECG Analysis Improves Diagnostic Accuracy and Reduces False STEMI Activations: A Multicenter U.S. Registry, she decreased false positive activations in LVH by 91%!! (from 44 to 4).
Patient 2: 55 year old, no past medical history, with exertional chest pain and syncope, EMS Code STEMI based on the prehospital ECG.
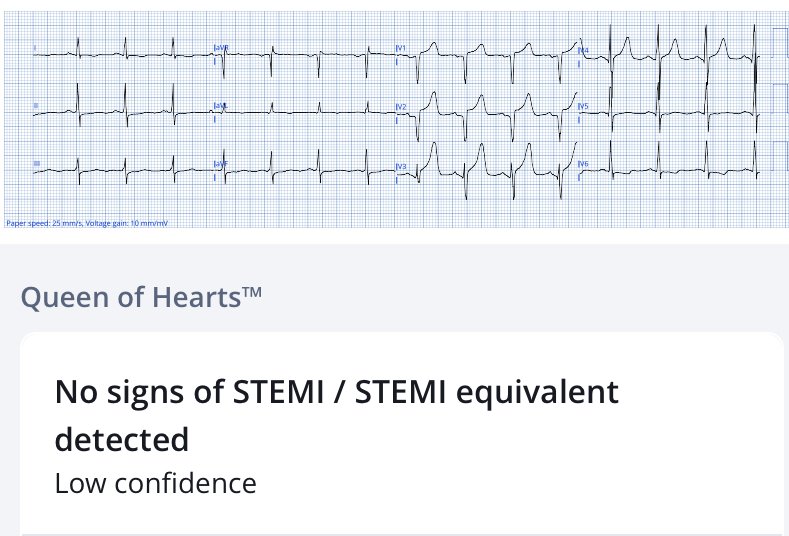
There’s voltage cutoff, which is very common in prehospital ECGs and this makes it difficult to assess proportionality, but there’s overlapping voltages in V4-5 and the morphology of ST segments is asymmetric. Despite the voltage cutoff the Queen finds no signs of OMI, but with low confidence.
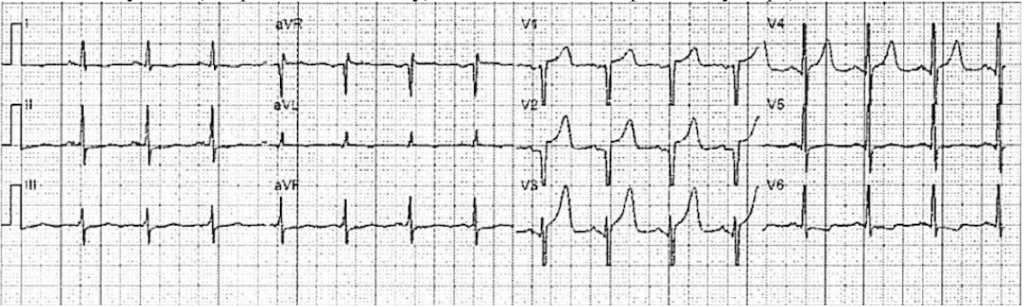
The ECG was repeated on arrival, revealing the full voltages, and now the Queen is confident there are no signs of OMI on the ECG
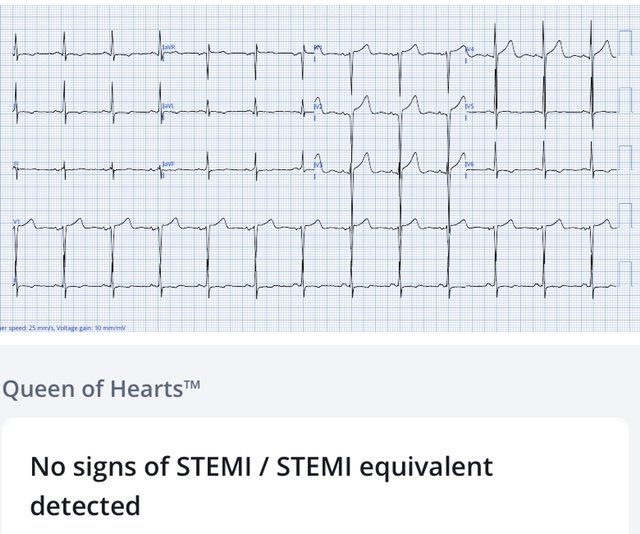
Cath lab was canceled and the patient was sent to the emergency department for assessment. Troponin rose from 30 to 55 ng/L, the patient was admitted as NSTEMI. Angiogram showed minimal coronary artery disease and echo showed severe aortic stenosis (explaining the exertional chest pain and syncope), treated with aortic valve replacement.
–
Patient 3: 60 year old, history of hypertension, with one week of chest pain and shortness of breath on exertion
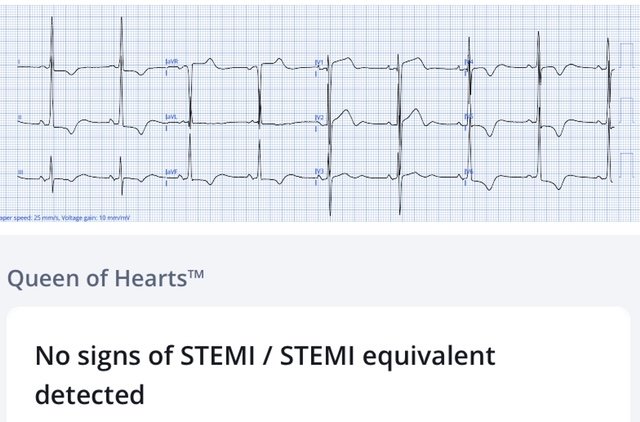
There are large and overlapping voltages, with discordant and proportional ST/T waves secondary to LVH. Cath lab was activated by the emergency physician but canceled by cardiology. Serial troponins were stable at 17ng/L (normal <16 in females) and echo diagnosed hypertrophic cardiomyopathy.
Patient 4: 55 year old, no past medical history, with 3 days exertional chest pain
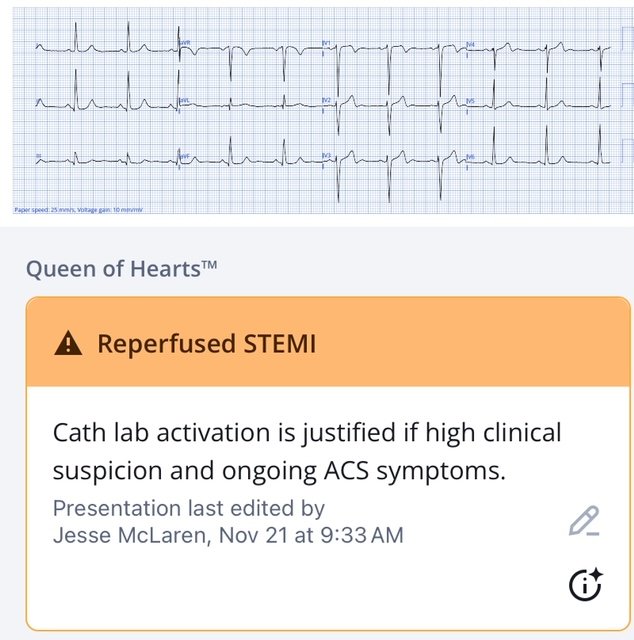
There’s minimal LVH criteria, making the biphasic T waves V2-4 concerning for primary ischemic changes from reperfusion (rather than secondary to LVH), identified by the Queen. Wellens syndrome requires resolution of ischemic symptoms and intact R waves (ie reperfusion before a major infarct); but V4 has lost most of its R wave, and when the first troponin returned at 8,000ng/L the nursing notes documented the patient still had chest pain.
But the patient was admitted as ‘high risk NSTEMI’, and after troponin rose to 12,000 two hours later the patient was taken to cath lab. Door-to-cath was 5hr40 minutes. There was 100% mid LAD occlusion, and peak troponin was 30,000 ng/L, with EF 48%. But because the patient was admitted as NSTEMI with delayed reperfusion, the discharge diagnosis remained NSTEMI despite a 100% occlusion. Next day ECG showed more obvious reperfusion T wave inversion:
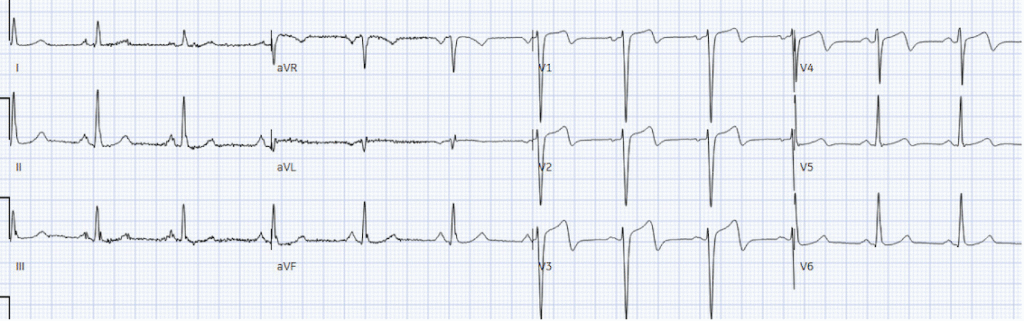
–
So four patients with chest pain, LVH on ECG and elevated troponin. How can we identify which had acute coronary occlusion:
- STEMI criteria: cannot be applied, so only 1 had a final interpretation of “STEMI” even though two had OMI. Technically, that one with STEMI was a STEMI(-) OMI, but had a final diagnosis of STEMI because there was a short door to balloon time.
- STEMI paradigm: in practice this led to one true positive STEMI activation, two false positive activations that were cancelled, and one false negative STEMI with delayed reperfusion. The true positive (patient 1) and false negative STEMI (patient 4) had the same angiographic finding of a 100% mid LAD occlusion, but the former had a discharge diagnosis of “STEMI” because of rapid reperfusion, while the latter had a discharge diagnosis of “NSTEMI” because of delayed reperfusion.
- OMI paradigm + AI: the Queen of Hearts, trained to interpret ECGs using totality and proportionality, accurately differentiated LVH with OMI vs LVH without.
Take away
- STEMI is defined as STE in the absence of LVH, so these criteria cannot differentiate LVH with or without OMI
- The first troponin can’t differentiate OMI from hypertensive heart disease
- LVH causes secondary, discordant and proportional ST/T changes – so using totality and proportionality can differentiate secondary repolarization changes from superimposed primary ischemic changes
- Queen of Hearts has been trained to identify LVH +/- OMI, which can reduce false positive activations and identify subtle occlusions
- Reperfusion requires both clinical and ECG evidence: resolved ischemic symptoms but ongoing ECG signs of occlusion, or reperfusion TWI with ongoing ischemic symptoms indicates ongoing occlusion
- Discharge diagnoses should be based on OMI vs NOMI (patient outcome), not STEMI vs NSTEMI (time to treatment)
Recent studies on Queen of Hearts:
-Baker P, Karim SR, Smith SW, et al. Artificial intelligence driven prehospital ECG interpretation for reduction of false positive emergent cardiac catheterization lab activations. A retrospective cohort study. Prehosp Emerg Care 2025;29(3):218-226
-Shroyer S, Mehta S, Thukral N, et al. Accuracy of cath lab activation decisions for STEM-equivalent and mimic ECGs: physicians vs AI (Queen of Hearts by PMcardio). Am J Emerg Med 2025 Nov;97:193-199
-Herman R, Mumma BE, Hoyne JD, et al. AI-enabled ECG analysis improves diagnostic accuracy and reduces false STEMI activations: a multicenter US registry. JACC Cardiovasc Interv 2025 Oct 15: S1936-8798(25):02600-7
= = =
======================================
MY Comment, by KEN GRAUER, MD (12/4/2025)
Excellent and insightful post by Dr. McLaren that proves its premise: Diagnosis of acute OMI ( = acute coronary occlusion) can be extremely difficult when there is marked LVH.
- The diagnosis of acute OMI with marked LVH becomes even more difficult — when there is also another form of significant heart disease. In such cases — deciding if to perform cardiac cath (and if so — then when to do so) becomes truly challenging.
= = =
CONFESSION: I had trouble interpreting these 4 initial tracings.
- Even when I immediately recognized in Patient #2 that QRS amplitudes were cut off in the pre-hospital initial tracing — I knew that I’d still be left with having to decide IF the very peaked anterior T waves were proportional to however deep the anterior S waves were — and even if so — still wondering what this example of marked LVH might be hiding within those huge QRST complexes.
- My “Take Home” Messages from Dr. McLaren’s post are the following: i) We should take comfort in knowing that the presence of marked LVH may make it difficult (if not impossible) to confidently include or exclude acute coronary occlusion on the basis of the initial ECG alone; — and, ii) Of great assistance in these problematic cases can be finding a prior ECG — and/or repeating the ECG in short order — serial Troponins — eliciting a more in-depth history — and especially bedside Echo (optimally done during chest pain — as this affords the greatest chance of finding a localizing wall motion abnormality).
- Applying my “Take Home” Messages to Patient #2 (who was found to have critical aortic stenosis in need of valve replacement) — the history of “exertional CP and syncope” provides a subtle initial clue, in that while these symptoms may clearly be associated with underlying coronary disease — they are less of a history of an acute event. Cardiac auscultation may have provided another clue (albeit the clinical reality that loudness and harshness of an AS murmur is not always indication of AS severity — and the murmur may be surprisingly “quiet” sometimes with severe AS in patients with reduced cardiac output). But higher-quality Echo would doubtless be suggestive of severe AS.
- In Patient #3 (who was found to have HCM = Hypertrophic CardioMyopathy) — Echo is absolutely diagnostic of HCM. This is fortunate — since although the ECG in most patients with HCM is not “normal”, as per My Comment in the October 28, 2023 post — ECG findings in patients with HCM are most often nonspecific (and non-diagnostic).
- In Patient #4 (who was in the midst of an extensive anterior infarction from acute LAD occlusion) — the initial Troponin = 8,000 ng/L immediately confirmed that the subtle terminal T wave inversions in leads V1-thru-V4 were “real” and given still ongoing CP, indicative of the ongoing extensive infarction.
= = =
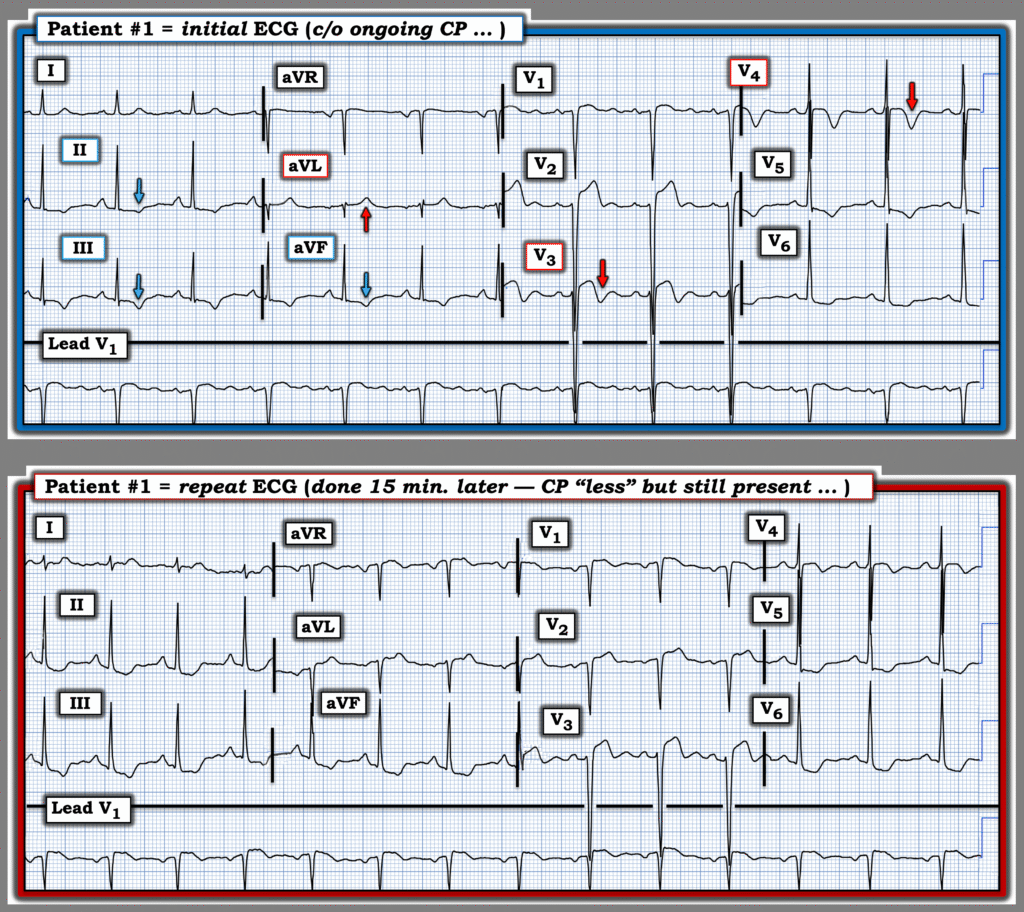
The 2 ECGs in Patient #1:
In Figure-1, to facilitate comparison between the initial and repeat ECGs from Patient #1 — I’ve placed these 2 tracings together.
- These 2 ECGs from Patient #1 illustrate that despite marked LVH — comparison with either a prior tracing or with a repeat ECG may show suspicious differences suggestive of an acute process.
- Confession: When I first looked separately at the initial ECG — and then at the repeat ECG — I did not appreciate the important differences between these 2 tracings. This emphasizes the importance of lead-to-lead comparison, without which it is too easy to overlook subtle-but-important changes.
- Note that there is a difference in the frontal plane axis between the 2 ECGs in Figure-1 ( = the QRS is small, but all upright in lead I in the initial ECG — but almost isoelectric in lead I of the repeat tracing). That said, while true that a change in frontal plane axis may produce some change in limb lead ST-T wave appearance — I thought the difference between the more peaked T waves (inverted in the inferior leads and upright in lead aVL) with minimal ST segment deviation — was clearly different than the increase in the amount of ST segment deviation and the change in ST-T wave shape evident in the repeat tracing.
- Similarly, in the chest leads — the depth of T wave inversion in leads V3,V4 was much greater in the initial ECG than in the repeat tracing. At the same time — the amount of J-point ST depression in leads V5,V6 is clearly greater in the repeat tracing.
- Bottom Line: In this patient reporting a change (reduction) in CP severity — I interpreted the above differences in ST-T wave morphology in at least 8/12 leads as indicative of “dynamic” ST-T wave changes — with need for prompt cath that confirmed acute LAD occlusion.
= = =
Figure-1: Comparison between the initial ECG and the repeat ECG from Patient #1. (To improve visualization — I’ve digitized the original ECG using PMcardio).
= = =
= = =