Written by Magnus Nossen
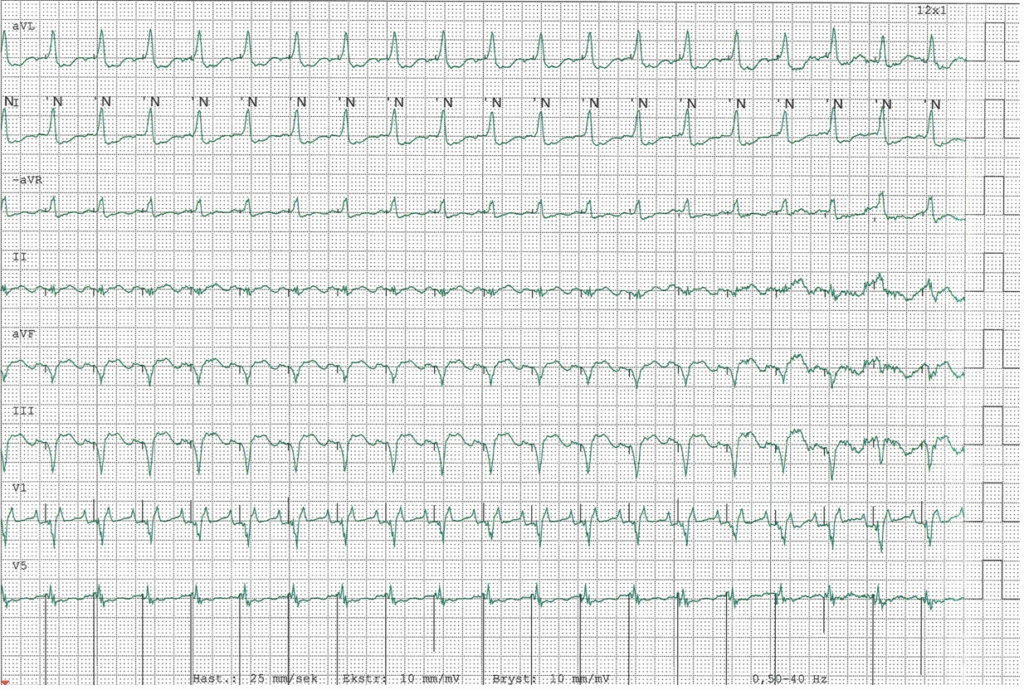
The patient in today’s case is an 87 year old female with a history of dual chamber pacemaker and Afib. She presented due to weakness and dyspnea that had progressed over a number of weeks. At presentation, the patient was tachycardic with a heart rate of about 120 BPM. Other vital signs were unremarkable. A prehospital ECG was transmitted and is shown below. What do you make of the ECG?
Prehospital ECG
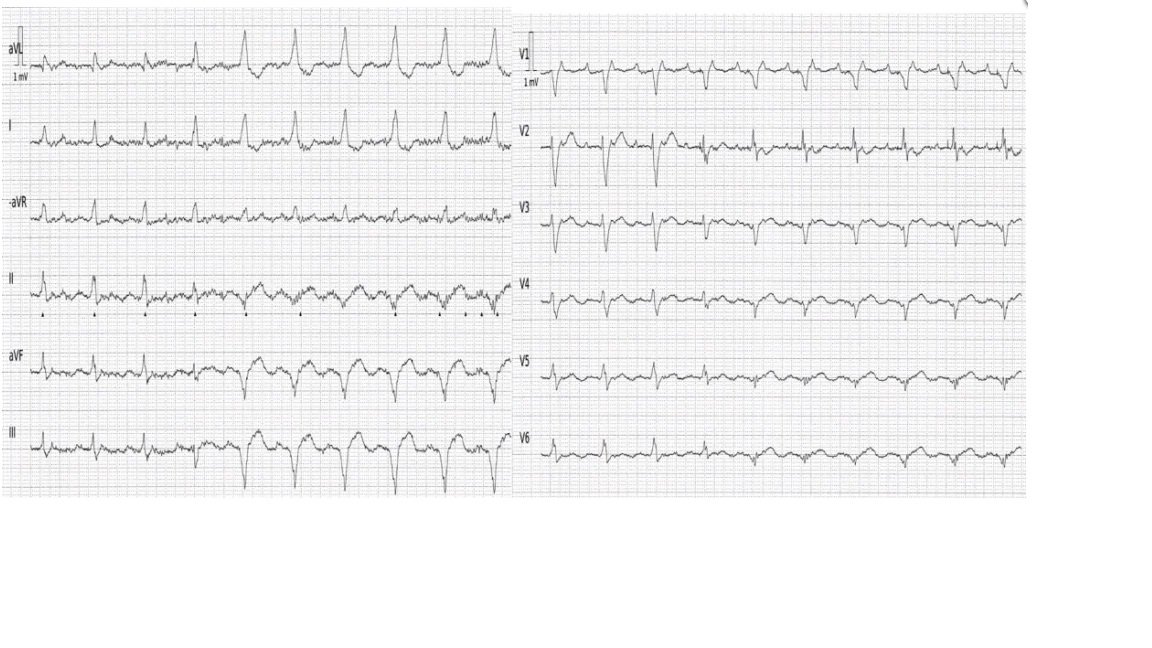
This ECG displays a regular rhythm featuring two distinct QRS morphologies. In the Cabrera format, the limb and precordial leads are recorded simultaneously. The first three beats show narrow QRS complexes, while the fourth beat exhibits a QRS morphology that is intermediate between the previous and the following complexes, which are wide. The QRS axis in the frontal plane is normal during the first three beats, but shifts to a superior and leftward axis thereafter.
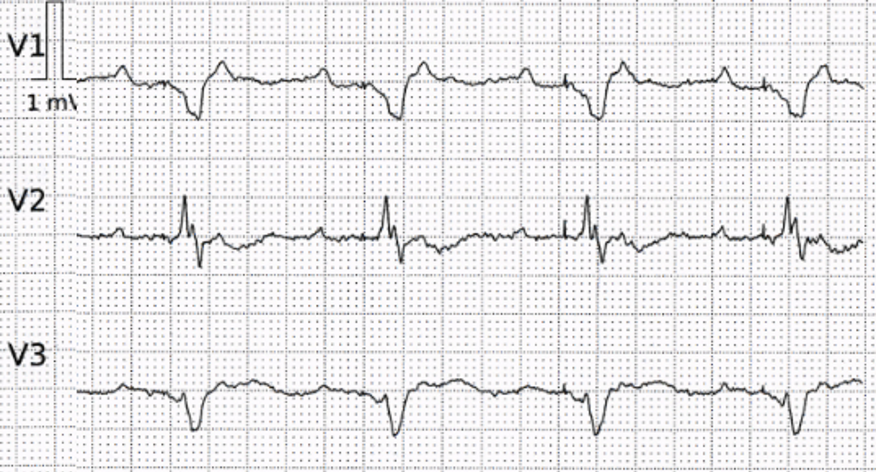
I am used to looking at ECGs in 50mm/s. Especially for rhythm interpretation, this faster paper speed can be beneficial. The image below shows the precordial leads in 50mm/s, and has better resolution/quality than the above compressed image. What is the rhythm and what accounts for the varying QRS morphology?
Precordial leads in 50mm/s
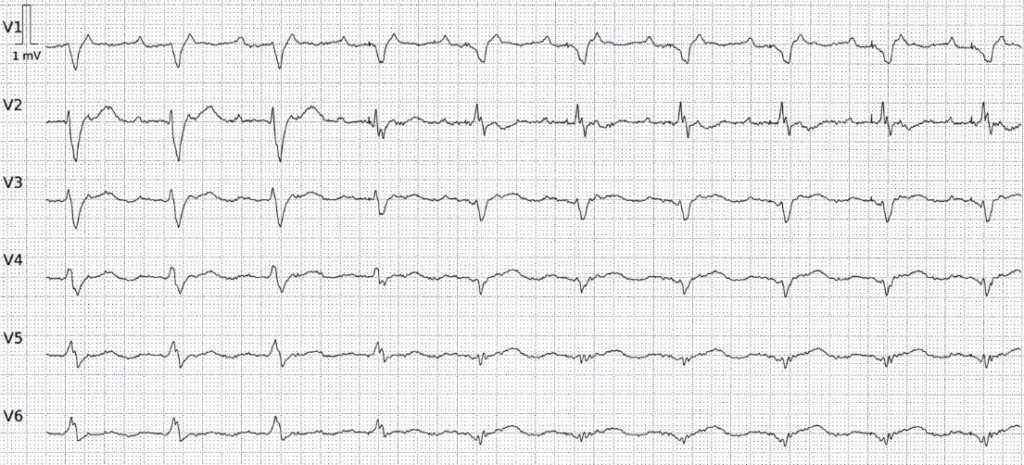
This ECG shows a regular atrial rhythm, with an atrial rate of about 240 beats per minute. The ventricular rate is half the atrial rate. So this is 2:1 atrial flutter. But what about beat #4? What accounts for the change in morphology of this beat and the susequent QRS complexes?
Let us have a closer look at the last QRS complexes in leads V1-V3. Below I have magnified these leads.
The orange markers encircle atrial actvity. Blue arrows are pointing to ventricular pacemaker spikes.
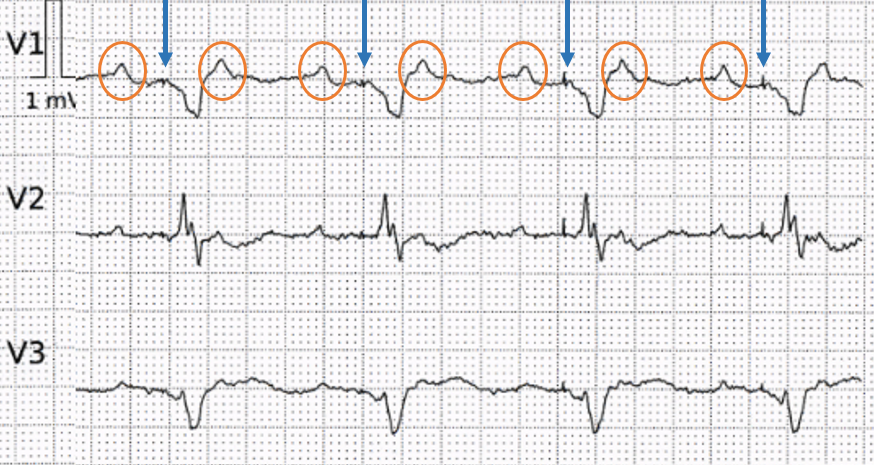
So — this ECG shows initial 2:1 conducted atrial flutter, and then subsequent ventricular pacing. This explains the axis shift noted on the initial ECG (superior and leftward compatible with right ventricular pacing). Beat #4 on the initial ECG is a fusion beat (ventricles partly depolarized intrinsically and by the pacemaker).
But why is the pacemaker pacing at a rate of 120/minute? Is the pacemaker malfunctioning? Is this pacemaker-mediated tachycardia?
See this case of Pacemaker Mediated Tachycardia. Ventricular Fibrillation, ICD, LBBB, QRS of 210 ms, Positive Smith Modified Sgarbossa Criteria, and Pacemaker-Mediated Tachycardia
No, this is not a typical case of pacemaker-mediated tachycardia (PMT), also known as endless loop tachycardia (ELT). In PMT, a ventricular paced event results in retrograde conduction through the AV node, producing an atrial depolarization. This retrograde P wave is then sensed by the pacemaker as though it was an intrinsic atrial event, which subsequently triggers another ventricular paced event followed by another retrograde P wave — creating a self-perpetuating “endless loop” of pacing. This phenomenon was more common with earlier generations of pacemakers. Modern devices now have effective algorithms that significantly reduce the risk of developing PMT.
What we observe in today’s tracing is functional 2:1 undersensing. This happens when the pacemaker “fails” to detect every other P-wave because the atrial activity occurs during the post-ventricular atrial blanking (PVAB) period.
Post ventricular atrial blanking (PVAB) refers to a pacemaker timing interval after a ventricular paced or sensed event in which the PM completely ignores signals on the atrial channel.
Post-ventricular atrial blanking (PVAB) is a feature in dual-chamber pacemakers designed to prevent the atrial sensing channel from detecting ventricular paced beats, far-field R waves, intrinsic ventricular events, and retrograde P waves. Both PVAB and the post-ventricular atrial refractory period (PVARP) are programmed timing intervals intended to reduce the risk of pacemaker-mediated tachycardia. The PVAB temporarily blocks the pacemaker from sensing any atrial signals immediately following a ventricular event, whether paced or sensed.
However, if an atrial depolarization from a tachyarrhythmia like atrial tachycardia or atrial flutter occurs during the PVAB, it will be undetected by the pacemaker. This leads to the pacemaker sensing only every other atrial event, a situation called 2:1 atrial blanking. For example, with an actual atrial rate of 240 beats per minute, 2:1 atrial blanking causes the pacemaker to perceive only 120 beats per minute, potentially resulting in ventricular pacing as if sinus tachycardia is present. This explains what happens in today’s case. The pacemaker “sees” sinus tachycardia and paces the ventricles at 120/minute.
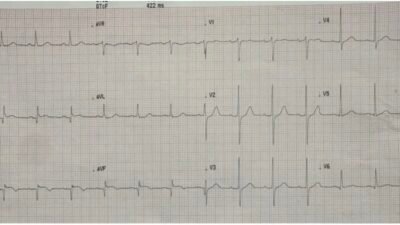
This observation was verified by pacemaker interrogation. The following screenshot from the pacemaker interrogation summary illustrates this phenomenon.
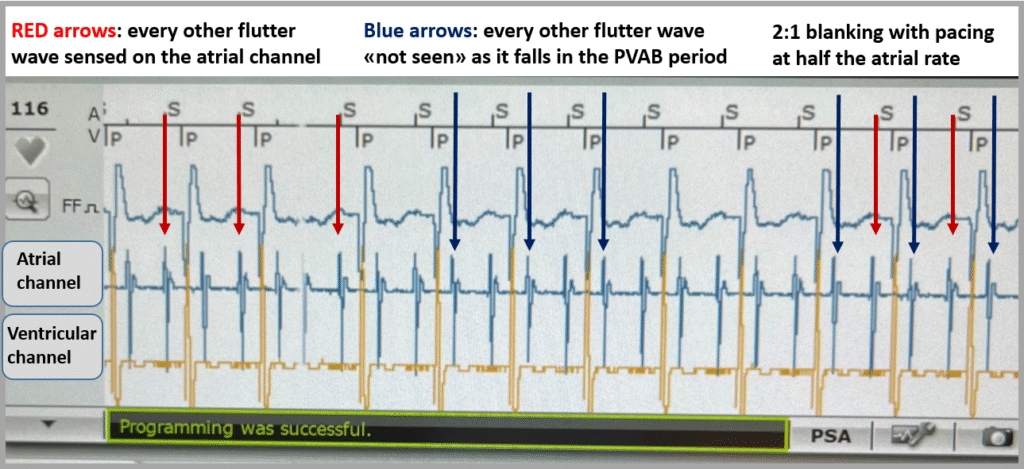
Telemetry print-out
Outcome
The pacemaker statistics showed that, since the last device interrogation, tachycardia, with a heart rate of about 120 beats per minute, was present approximately 75% of the time with ventricular pacing occuring for more than 80% of the ventricular beats. Thus — 2:1 blanking was a significant issue for this patient. The pacemaker was reprogrammed to VVIR mode at 70 beats per minute. In VVI mode, the pacemaker will ignore the atrial channel all together (and will therefore not track any atrial activity). This programming feature in effect turns a two-chamber pacemaker into a single chamber ventricular pacemaker. In today’s patient, this change did not immediately affect the heart rate, as the patient continued to have 2:1 blocked atrial flutter due to intrinsic 2:1 AV block.
After increasing the beta-blocker dose, a higher degree of AV block was achieved, and the the ventricular rate was well controlled. If the beta-blocker dose had been increased without adjusting the pacemaker settings — it is likely that this patient would have continued to be paced at half the atrial rate due to 2:1 blanking. This would have put her at risk of both tachycardia-mediated and pacing-induced heart failure.
A different management strategy would have been appropriate if the patient had been younger with fewer comorbidities. In such cases, rhythm control with cardioversion followed by flutter ablation is a better option. However, this patient was very elderly and frail, with echocardiography revealing marked dilation of both the right and left atria; these factors made rate control the more suitable therapeutic approach.
= = =
======================================
MY Comment, by KEN GRAUER, MD (12/3/2025):
Credit to Dr Nossen for his explanation in today’s post of PVAB (Post-Ventricular Atrial Blanking) — which is just one of an ever increasing number of sophisticated pacemaker concepts that these amazing devices are capable of. Gone are the days when it was easy to determine if the pacemaker is (or is not) working as it should. Fortunately — the option to consult with cardiology for formal pacemaker interrogation is now readily available to provide a definite answer.
- Many of us (myself included) are not expert in pacemaker troubleshooting (beyond an awareness of the often forgotten but highly useful truism that most of the time — the pacer is right!). As a result — most of the time, our principal task is to figure out why despite whatever unusual ECG findings are present — more likely than not, the pacemaker is functioning correctly.
- As emphasized by Dr. Nossen — even the previous nemesis of pacemakers ( = Pacemaker-Mediated Tachycardia) has been largely eliminated by sophisticated algorithms designed to counter the retrograde atrial conduction that so often precipitated PMT in the past.
= = =
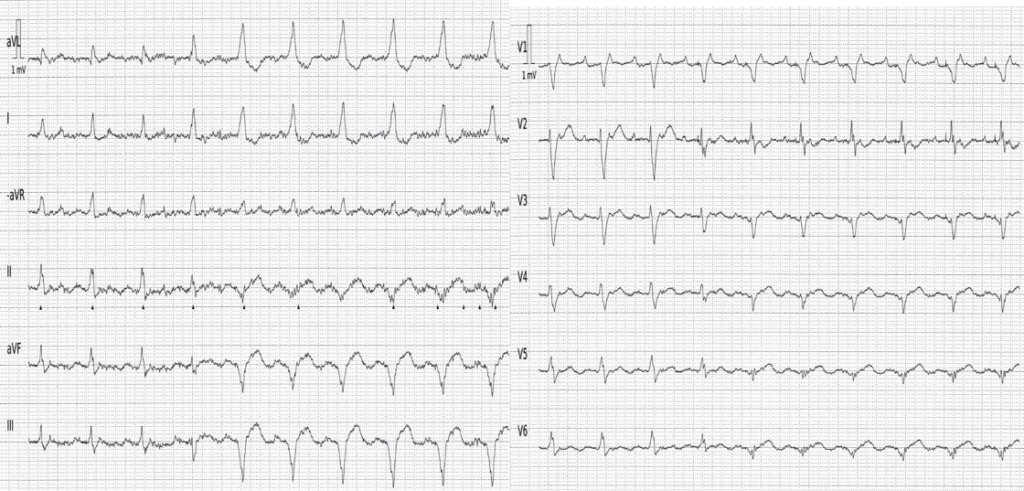
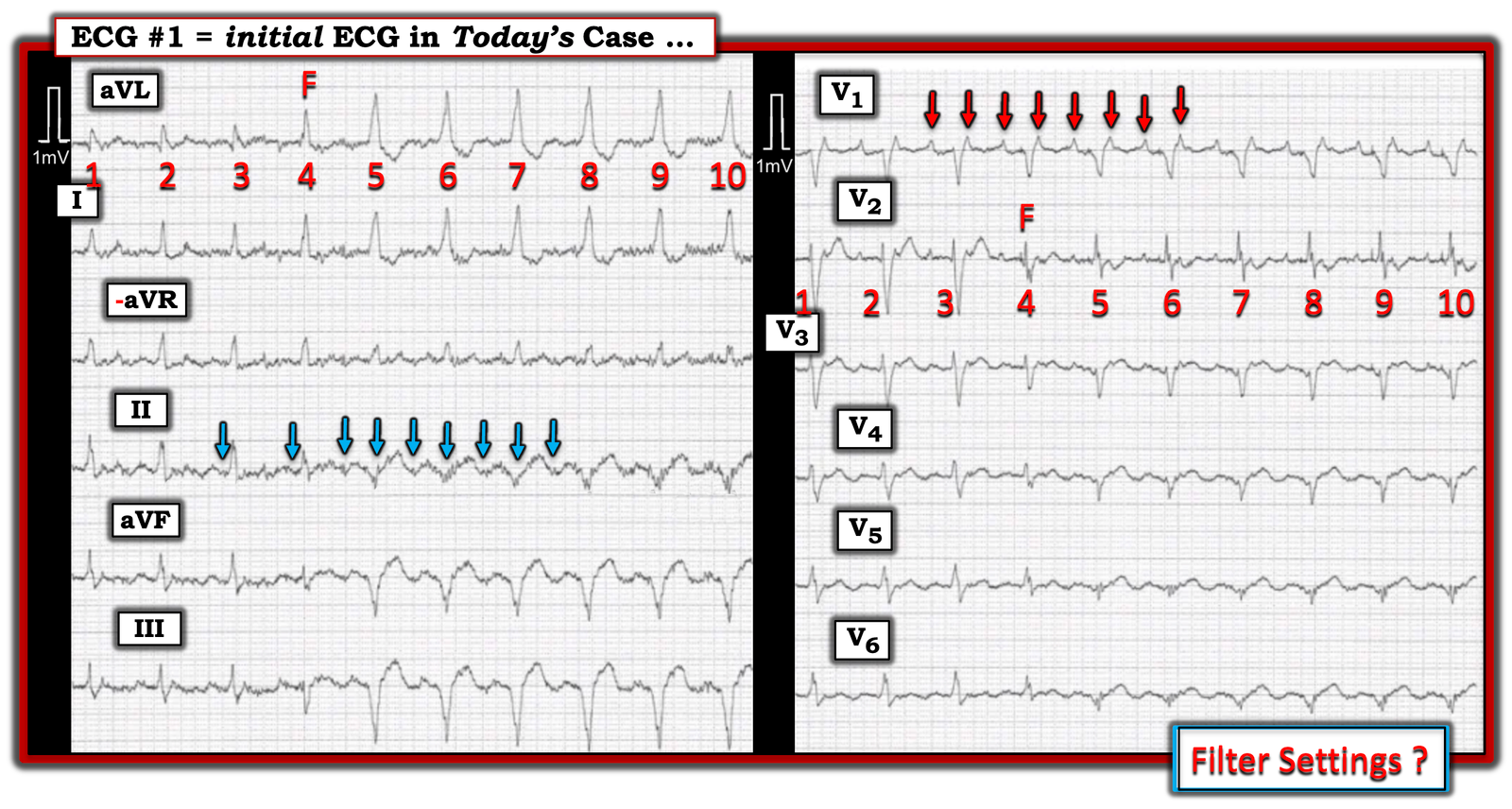
Today’s Initial Tracing:
And so I found myself uncertain about what was going on with today’s initial ECG (that for clarity — I’ve reproduced and labeled in Figure-1). We are told that today’s 87-year old female patient had a history of AFib with implantation of a dual-chamber pacemaker for some unknown indication.
QUESTIONS:
- If this patient has a pacemaker — Why can’t we see pacemaker spikes on the initial ECG?
- And, if the patient does have a pacemaker — Is the pacemaker functioning correctly?
= = =
Figure-1: I’ve labeled the initial ECG in today’s case.
MY Thoughts:
I would not have known from looking at Figure-1 that today’s patient has a permanent pacemaker — because I simply do not see pacemaker spikes.
- Filter settings are not displayed on ECG #1. To emphasize — it may be that the reason there is no indication of filter settings used for recording this tracing is that notation of filter settings was removed during the process of compressing this ECG that was originally recorded at 50 mm/sec. Regardless — the point is that all-too-often the potential effect that a given filter setting might have on the ECG feature you are seeking to optimize is ignored!
- We’ve periodically reviewed the potential role of filter settings in Dr. Smith’s ECG Blog (See My Comment in the March 4, 2019 post, among others). Pacing spikes are a high frequecy signal. As a result pacing spikes may be filtered out (and therefore not seen on the ECG) if the low pass filter is set at too low of a value (ie, below 150 Hz). In this case — a wide QRS rhythm without P waves may be seen (and may easily be misinterpreted as either a junctional rhythm with an LBBB pattern or as a ventricular rhythm).
= = =
So, What is Going On in ECG #1?
As per Dr. Nossen — the same 10 beats are seen in both limb leads and chest leads. I found awareness of this fact helpful to appreciate — because no single lead provided all the answers — such that seeing all 10 beats in each of the 12 leads allowed me to optimally reason out various relationships.
- As was Dr. Nossen — my “eye” was immediately drawn to the presence of 2 distinctly different QRS morphologies (with this being best seen in lead aVL). The QRS appears to be narrow for the first 3 beats in the tracing. The QRS widens for beats #5-thru-10.
- As per Dr Nossen — beat #4 manifests a QRS and T wave morphology that is intermediate between the first 3 narrow beats and the last 6 wider beats. Therefore, beat #4 is a fusion beat (“F“). But — What is it that is “fusing”?
- Significant baseline artifact (especially in the limb leads) — makes determination of atrial activity difficult. As a result — my “eye” was drawn to lead V1, as the best lead in ECG #1 for assessing atrial activity. RED arrows in this lead reveal 2:1 atrial activity (ie, The underlying rhythm is Atrial Flutter, with flutter waves at a rate of ~240/minute).
- Recognizing regular flutter activity in lead V1 — allowed me to go back and take another look at potential atrial activity in the limb leads. As per the BLUE arrows — I believe that I now do see regular, 2:1 atrial activity — at least for a portion of lead II (albeit baseline artifact had just a moment ago suggested to me that atrial activity was not consistent throughout this 10-beat rhythm strip).
- KEY Point: I carefully measured all intervals with calipers. All intervals remain constant throughout the tracing. Despite the change in QRS morphology — the R-R interval does not change over the course of this 10-beat rhythm strip. In the one lead in which 2:1 atrial activity is clearly present ( = lead V1) — both the P-P interval and the PR interval preceding each QRS complex remain constant. This would not be the case if these wide beats were of ventricular etiology. Therefore, despite my inability to convince myself that there are pacing spikes — the fusion beat and widening of the QRS for beats #5-thru-10 has to be the result of paced QRS complexes.
- BOTTOM Line: Although I was unaware of the concept of PVAB prior to reading Dr. Nossen’s discussion — my above “thought process” allowed me to strongly suspect that widening of the QRS (that begins with beat #5) — but without any change in R-R, P-P and PR intervals — had to be indication of pacemaker activity, with probable appropriate pacemaker function given maintenance of a consistent (albeit rapid) ventricular rate.
- Fortunately — formal pacemaker interrogation in today’s case was able to confirm appropriate pacemaker function. An increase in the ß-blocker dose, together with pacemaker reprogramming to VVIR mode restored an appropriate ventricular rate.
- As is so often the case — the pacemaker was doing precisely what its ingenious programming designed it to do.
= = =
= = =