Written by Pendell Meyers
A man in his 60s with history of prior CAD with PCI, HTN, HLD, and aortic insufficiency presented with acute chest pain radiating to the left arm. He had been walking on a treadmill for approximately 5 minutes when the symptoms began abruptly. He had associated shortness of breath, diaphoresis, and dizziness.
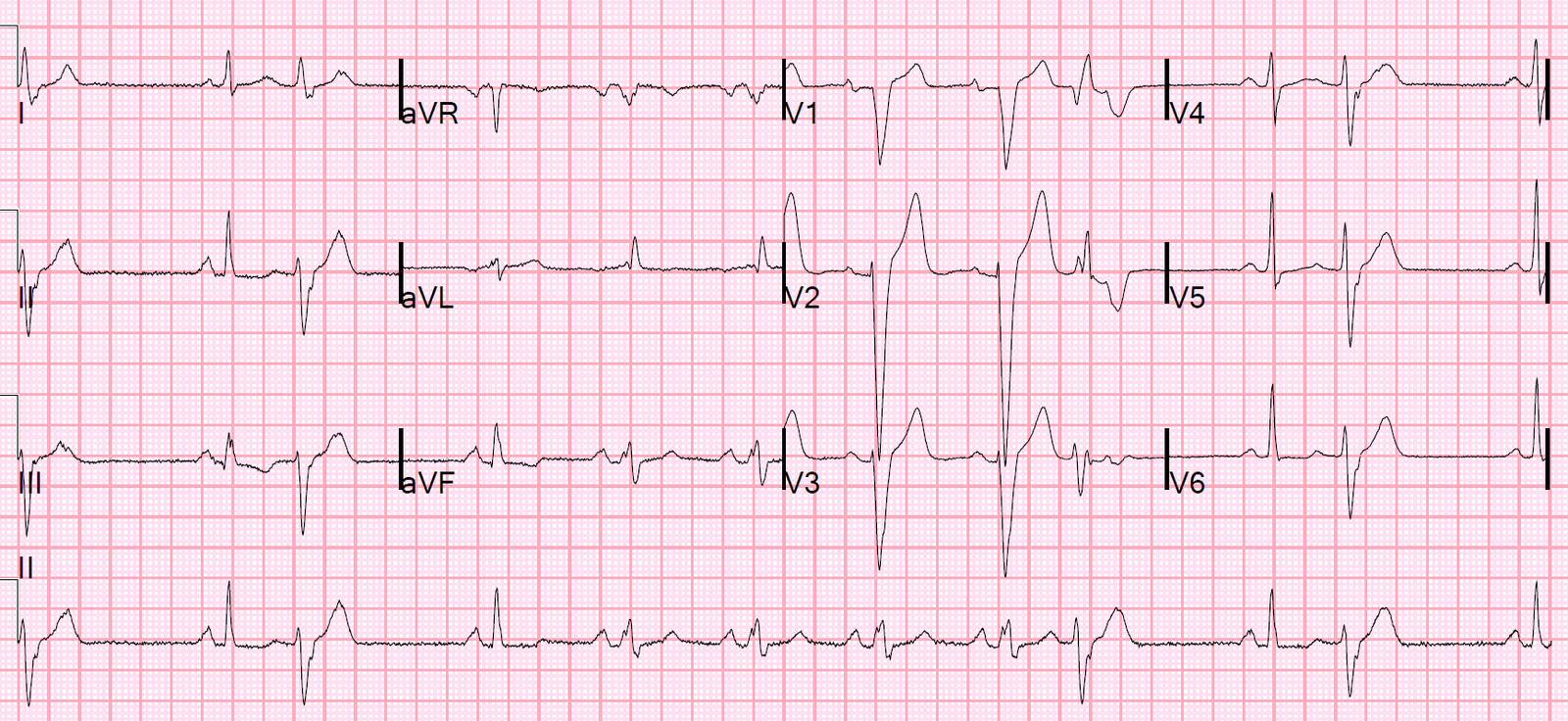
He arrived by private vehicle with relatively normal vitals signs and this initial ED ECG:
 |
| What do you think about the patients clinical presentation? About the ECG? |
The patient: the treating team was completely convinced of ACS (or less likely another catastrophe such as dissection) by his symptoms and clinical appearance before the ECG was recorded.
The ECG read prospectively: Both I and Dr. Smith read this ECG (without any clinical information) as having no convincing evidence of OMI. We both noted the slight STE in V1 along with minimal STD in V6, I, and aVL, stating that these findings were possibly concerning and that we would perform serial ECGs as well as clinical info, echo, etc, to help the decisions further. We have described the pattern of STE in V1-V2 with lateral STD in cases of LAD occlusion, but we both agreed that something about this ECG just doesn’t fit that pattern. But overall with this ECG in isolation (no clinical context, no baseline available to us at that time), we did not see evidence of OMI.
Smith comment: There is a touch of STE in V2 and V3, so one might wonder if this is subtle LAD occlusion. The formula value (using QTc of 400) is 16.88 however, which is very low and makes it very unlikely that this STE is due to LAD occlusion (which is not saying the same thing as there is unlikely to be LAD occlusion – sometimes it just does not show on the ECG).
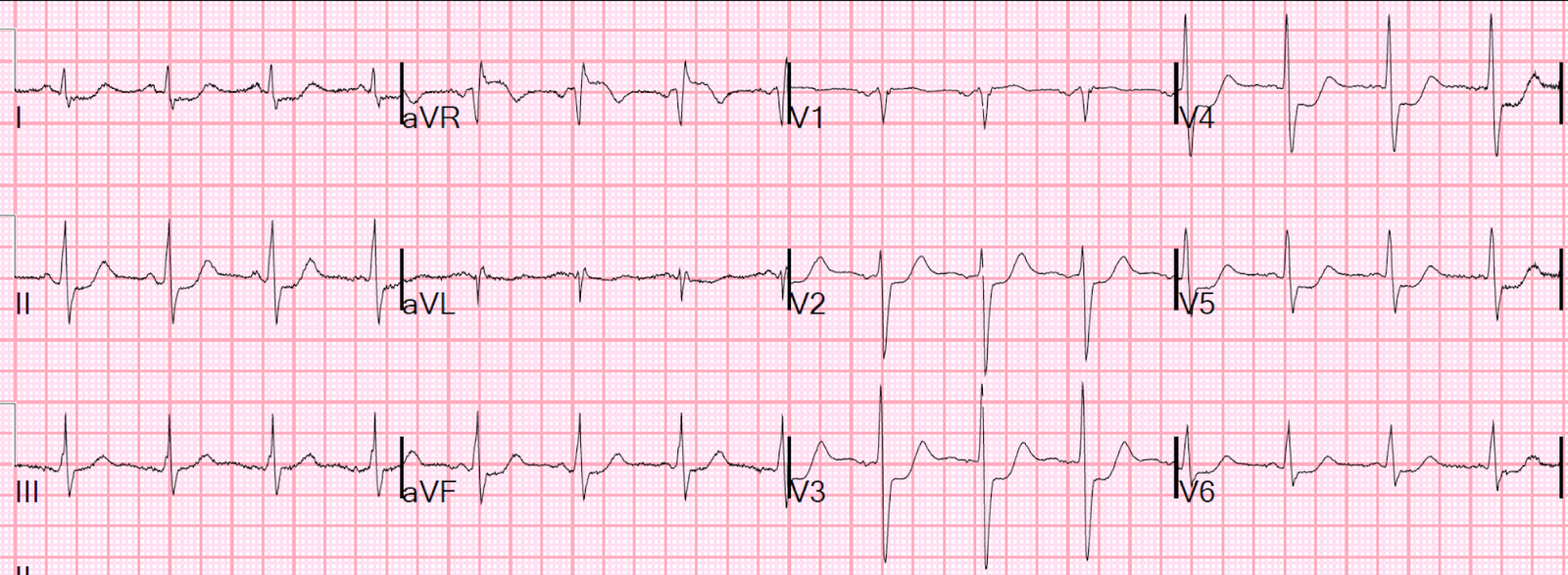
There was a baseline ECG on file that they found later:
 |
| Baseline ECG which is basically normal. |
Given ongoing ischemic pain and clinical appearance and pretest probability, the treating team called a “heart alert” (which summons the cardiology fellow to bedside immediately and coordinates a phone call between ED and Cardiology interventional attendings, to discuss potential activation of the cath lab).
Another management option might include recording both right sided and posterior leads. Sometimes when the standard 12-lead shows no evidence of OMI, the RV and/or posterior wall will manifest some ST elevation. It is worth a try.
Luckily, the cardiology attending was already in the building and came immediately to evaluate this patient whom he knew well.
They reviewed the case at bedside and agreed that his clinical appearance and history warranted emergent cath lab activation regardless of the lack of clear ECG findings, and before the first troponin would return.
The team performed a bedside echo which is not available, but they could not see any clear wall motion abnormality.
These two ECGs were recorded before he left the ED to the lab:
In the lab he was found to have complete occlusion of the proximal RCA (see images below). This was stented with excellent angiographic result.
 |
| Before intervention. Total proximal RCA occlusion. |
 |
| After intervention, showing the course of the RCA. |
Post cath ECGs:
 |
| This shows small terminal T-wave inversions which are indicative of inferior reperfusion in leads III ad aVF. |
The patient did well and was discharged home.
In retrospect, the only findings of proximal RCA occlusion that I can clearly see on the pre-cath ECGs is STD in aVL and lead I (which are minimal and obscured by baseline wander).
Learning Points:
The ECG is quite accurate overall when interpreted by those with significant experience. However, it will never be enough in isolation to detect all acute coronary occlusions. This is one of the reasons why the name of the paradigm designed to find acute coronary occlusion must be centered on the pathology we are trying to find, and must not contain specific ECG findings (such as “ST Elevation MI”).
When you instead understand the better name “Occlusion MI,” then you give your mind permission to keep considering the possibility of acute coronary occlusion despite the lack of traditional ECG findings, and your mind is free to consider other clinical features that may help with this important diagnosis such as clinical presentation, persistent pain, echo findings, etc.
Take it from someone who cares a lot about ECGs – it’s not all about the ECG!
This case is an excellent example of the “no false negative paradox.” This paradox is one of the major problems with the current STEMI paradigm, and it describes the situation that there is no such thing as a “false negative” in the STEMI paradigm. For every other paradigm, there is an actual outcome that defines whether the patient had the underlying disease or not, whether the case was a true negative or a false negative (for example, if you decide that a patient does not have appendicitis based on your history and physical exam findings, and the patient later turns out to have appendicitis, then this case would be classified as a false negative). But in the STEMI paradigm, the definition of the paradigm is a millimeter finding on a piece of paper, and has nothing to do with the patient’s actual outcome(!), and there can be no false negative because NSTEMI by itself does not warrant emergent management by definition of the paradigm. Thus, even a patient who dies of acute coronary occlusion is not considered a “false negative” in the STEMI vs. NSTEMI paradigm, so long as their ECG doesn’t manifest the specific criteria (which have pitiful accuracy). This case is an excellent example of a false negative in the OMI vs. NOMI paradigm, which cannot exist in the STEMI vs. NSTEMI paradigm. For more discussion of the no false negative paradox, hear the discussion in this lecture: https://emcrit.org/emcrit/emcrit-podcast-250-the-omi-manifesto-lecture-by-pendell-meyers/
===================================
MY Comment by KEN GRAUER, MD (4/20/2020):
===================================
Highly insightful case by Dr. Meyers — because (as Dr. Meyers emphasizes) — the ECG shows very little despite cath confirmation of acute proximal RCA occlusion.
- Cases like this are humbling. I think they teach us a lot!
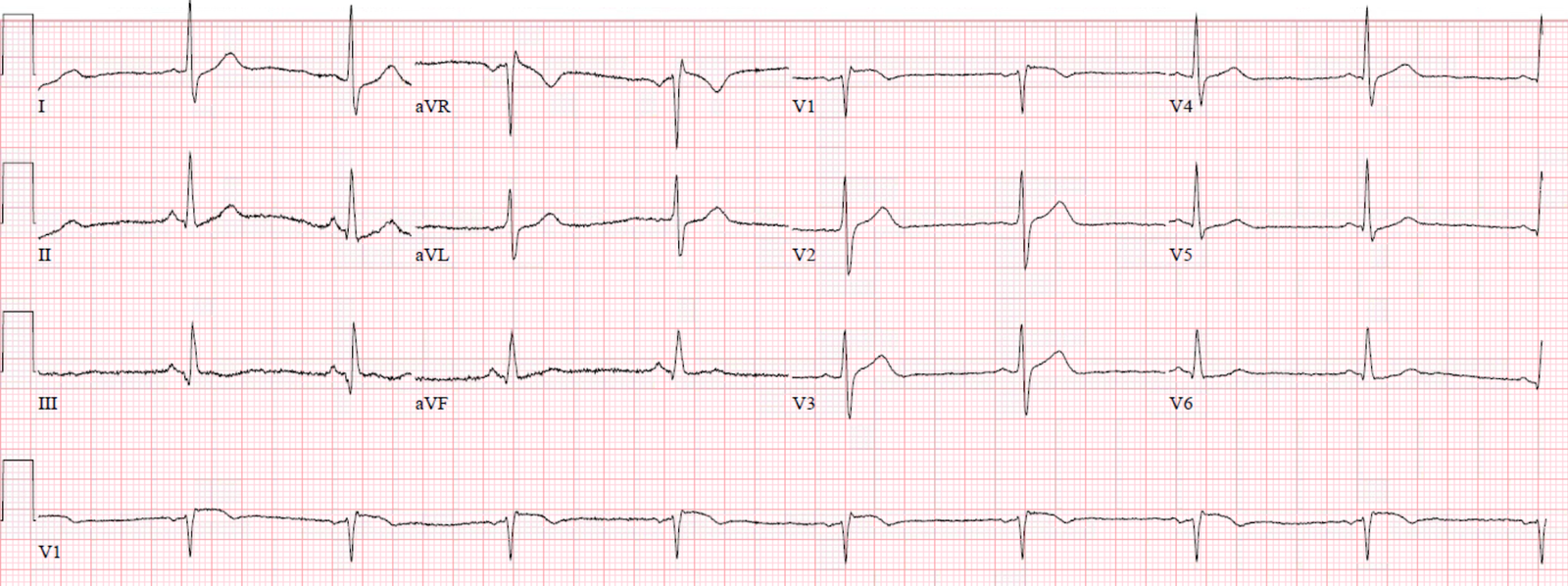
For illustrative purposes — I have put in Figure-1 the initial ECG that was done in the ED ( = ECG #1) — together with a baseline tracing ( = ECG #2) that was previously done on this patient.
- Given the “high prevalence” situation in this case (ie, the patient is a man in his 60s with known coronary disease and new-onset worrisome chest pain!) — the onus falls on us to rule out an acute event, rather than the other way around. I therefore scrutinized ECG #1 for the smallest of details.
 |
| Figure-1: The initial ECG in this case — compared to a prior tracing (See text). |
MY THOUGHTS on ECG #1: I agree completely with Drs. Meyers and Smith that ECG #1 does not manifest convincing evidence of OMI.
- There is slight sinus arrhythmia and bradycardia (rate just under 60/minute). All intervals and the axis are normal. There is no chamber enlargement. There are small inferolateral q waves. Perhaps the inferior Q waves reflected a prior event in this patient with known coronary disease (?) — but there certainly were no acute inferior lead changes. ST-T waves in leads III and aVF, as well as in the lateral chest leads were a bit flattened — and the ST segment in lead aVL was straight — but these were clearly nonspecific changes.
- Transition occurs early — in the form of a surprisingly tall R wave in lead V2 (R=S in this lead). Perhaps this reflected posterior infarction at some point in time — but again, nothing that looked acute.
- There is some subtle straightening of the ST segment in lead V3 — and the J-point in leads V2 and V3 appears to be slightly elevated. That said — I was not convinced this was acute.
- What did catch my eye — was the clearly abnormal ST elevation in lead V1. I did not know if this was ischemic or a technical mishap. The fact that there was actually an rSr’ complex in lead V1 of ECG #1, with a similar QRST appearance in lead aVR raised the possibility of too-high placement of the lead V1 electrode on the chest.
- BOTTOM LINE: In a “high prevalence” situation in this patient who presented with worrisome new-onset chest pain — I thought the ST-T wave appearance in lead V1 of ECG #1 was clearly out of the ordinary. More information (serial tracings, stat Echo, troponin — and in this case, prompt cardiac cath) was in order.
ECG #2 (the Baseline Tracing) — Although there is slight difference in the frontal plane axis, and in QRS morphology of some chest leads in ECG #2 compared to ECG #1 — none of these slight differences looked acute.
- The one lead that I thought did show a potentially significant change since the baseline ECG was done was lead V1. The ST-T wave in lead V1 of the baseline ECG ( = ECG #2) looked normal. In contrast — there was unexpected ST elevation in lead V1 on the initial ED tracing ( = ECG #1). That said, the terminal r’ was not present on the baseline tracing — which suggested the reason for ST elevation in lead V1 of ECG #1 might simply be the result of lead misplacement of the V1 electrode. (For more on recognizing lead misplacement of leads V1,V2 — Please see My Comment in the November 4, 2018 post in Dr. Smith’s ECG Blog).
- Alas — nothing evolved on serial ECGs done prior to cath. None of these follow-up tracings showed abnormal ST elevation or a terminal r’ in lead V1.
Last Lesson — I learned from this case by going back through the serial ECGs that were done.
- I thought it important to recognize in this patient with worrisome new-onset chest pain — that the ST elevation we see in lead V1 of ECG #1 is not a usual finding. Whether the reason for this ST elevation was lead misplacement of the V1 electrode and/or transient ST elevation from acute proximal RCA occlusion (that was not seen on subsequent pre-cath ECGs) — I think is uncertain.
- Finally — I found it insightful in this case that serial ECG tracings showed very little despite cath confirmation of acute proximal RCA occlusion.
- P.S. — I would have been curious to see what right-sided leads done at the time ECG #1 was recorded would have looked like.
Our THANKS to Dr. Meyers for presenting this case.