By Willy Frick
CASE PRESENTATION
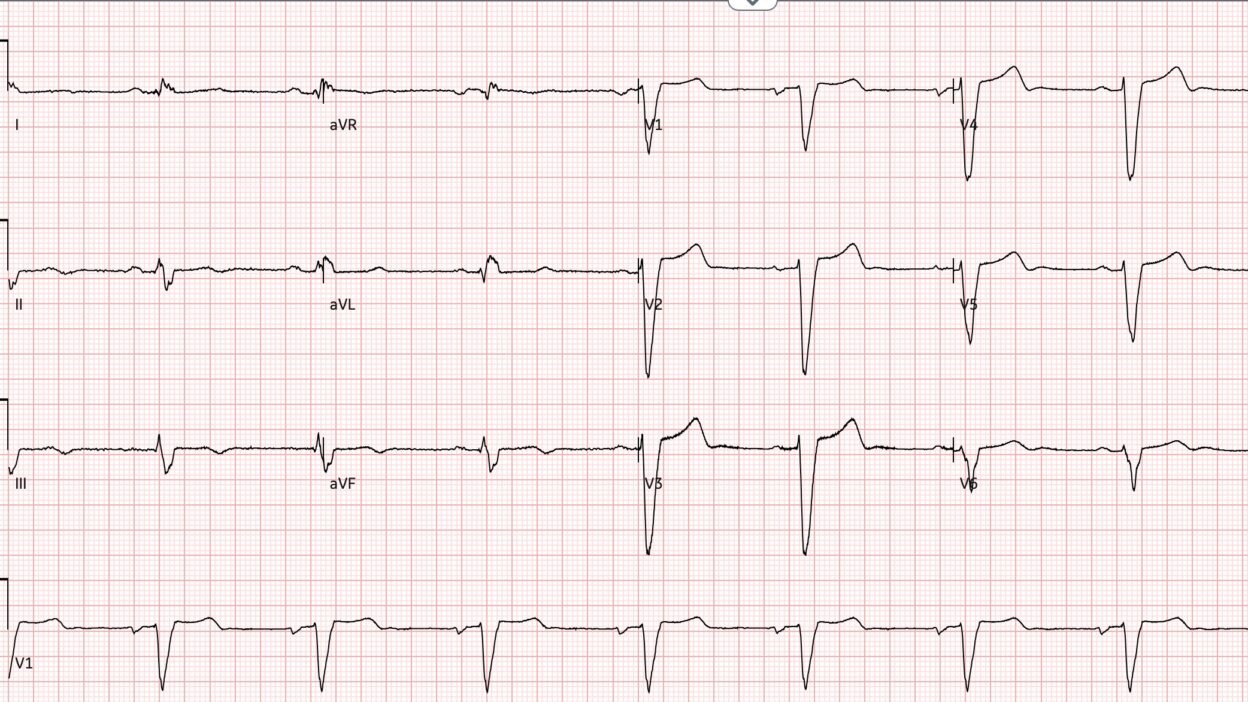
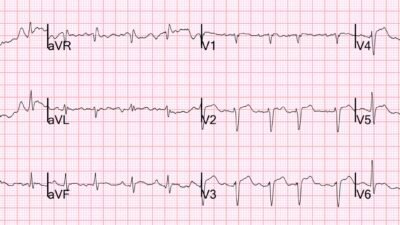
A middle aged woman with HFrEF (EF 25-30%) and persistent atrial fibrillation was admitted to the hospital electively for loading with dofetilide. Dofetilide is a class 3 anti-arrhythmic drug which is useful in patients pursuing a rhythm control strategy for atrial fibrillation. A rhythm control strategy is an attempt to try to restore sinus rhythm. Dofetilide must be initiated in the inpatient setting due to the known risk for Torsades de Pointes (TdP). The risk of TdP is particularly elevated with a low ejection fraction, as in this case. The patient’s baseline ECG is the first one shown below.
Smith Comment (relevant especially for Emergency Physicians): Dofetilide is the same class of drug as ibutilide, which can be used to facilitate cardioversion of atrial fibrillation in the ED (see detailed and important discussion at the bottom of the post). Dofetilide and ibutilide are Class III antiarrhythmic agents. They function as potassium channel blockers, primarily inhibiting the rapid component of the delayed rectifier potassium current. This is the current that repolarizes the myocardium. If you delay that current, you delay repolarization, and thus prolong repolarization and thus prolong the QT interval. So the primary complication of dofetilde and ibutilide is prolonged QT and risk of Torsades de Pointes.
Figure 1: Baseline ECG
Here the heart rate is 55.
The machine calls the QT 526 ms, which appears to be correct and is on the long side. However, there is a little more leniency in patients with wide QRS. Strictly speaking, the label does not recommend initiation of dofetilide in patients with wide QRS and QTc (or uncorrected QT for heart rates under 60). However, there is practice variation on this point and many electrophysiologists practice discretion based on the absolute width of the QRS.
Smith: note there is also sinus bradycardia. This makes the likelihood of TdP substantially higher because it allows for a longer refractory period and higher likelihood of a PVC. This is otherwise known as “Pause dependence” as any pause increases the likelihood of TdP.
Why is this?
The risk of TdP is proportional to QTc. TdP occurs when a PVC depolarizes the ventricle when it is still partially refractory. This is called the “R on T phenomenon.” The PVC is an R wave, and the T wave is the vulnerable period when the ventricle is partially refractory. It helps to review this image from Life In The Fast Lane:
Figure 2: Cardiac Intervals
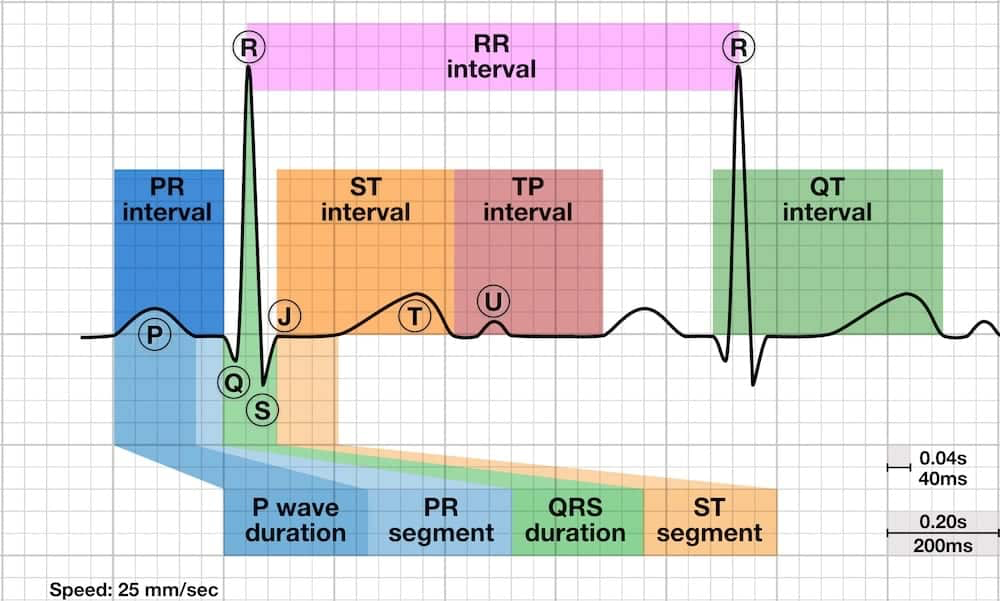
Here, we clearly see that:
QT interval = QRS duration + ST interval
Therefore, anything the prolongs the QRS duration (for example intraventricular conduction delay in our patient’s case) will also necessarily prolong the QT interval. But it is really just a portion of the ST interval that constitutes the vulnerable period for R on T. During the QRS, the ventricle is already depolarizing. If a part of the ventricle were stimulated VERY soon after the QRS, the ventricle would be absolutely refractory, and nothing would happen. But if stimulation occurred a little bit later (somewhere during the middle to end of the T wave), the ventricle is only partially refractory and can depolarize again leading to TdP.
Smith: this is why some authors state that the best way to measure QT prolongation in left bundle branch block (LBBB) is to use the T-peak to T-end time (TpTe). That is the interval in which there is the highest risk and the interval which is lengthened in long QT. A prolonged TpTe time is between 85-100ms. See our article: Dodd KW, Elm KD, Dodd EM, Smith SW. Among patients with left bundle branch block, T-wave peak to T-wave end time is prolonged in the presence of acute coronary occlusion. Int J Cardiol [Internet] 2017;236:1–4. Available from: http://dx.doi.org/10.1016/j.ijcard.2017.01.064
Some authors recommend Rautaharju correction for wide QRS.
——QTcR = QT – 0.185(RR – 1) + k, where k = +0.006 for men and 0 for women.
Others recommend using the JT interval and corrected JT interval (JTc).
——(JTc = QTc – QRSd)
Back to the case:
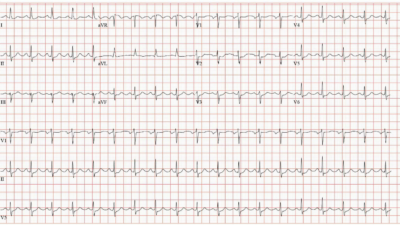
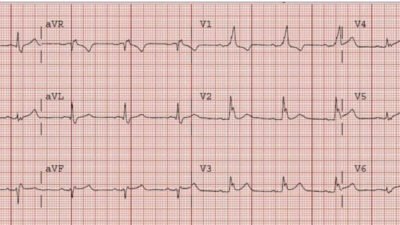
Out of an abundance of caution, her clinical team chose to start her at 250 mcg. (The usual starting dose for a patient with normal renal function is 500 mcg.) The ECG 2 hours after this dose is shown below:
Figure 3: ECG 2 hours after first dose of dofetilide
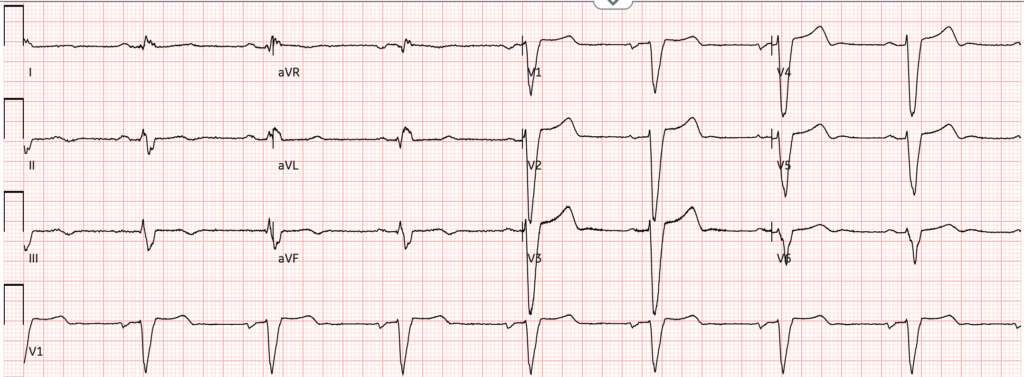
The heart rate is dangerously slow at 48 bpm. QT has increased to about 560 ms, significantly longer than the previous 526 ms.
If the QTc (or QT if HR < 60) rises above 550 ms in patients with a wide QRS, the dose should be reduced by 50%. The QTc here was measured at 554 ms. The order was changed so that the following morning, the patient would receive a dose of 125 mcg.
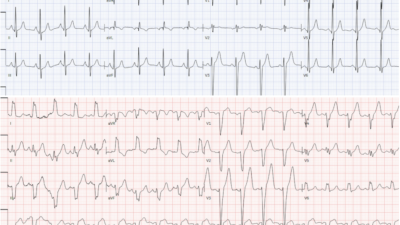
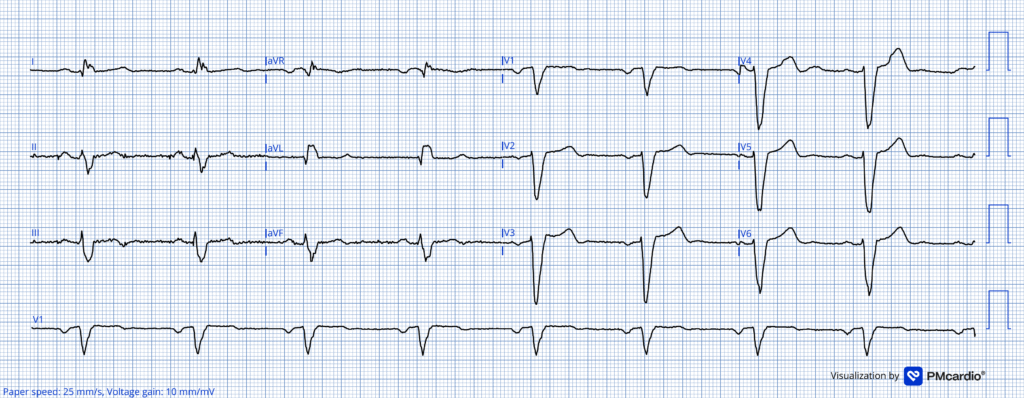
However, later that evening, telemetry showed the following:
Figure 4: Cardiac Telemetry
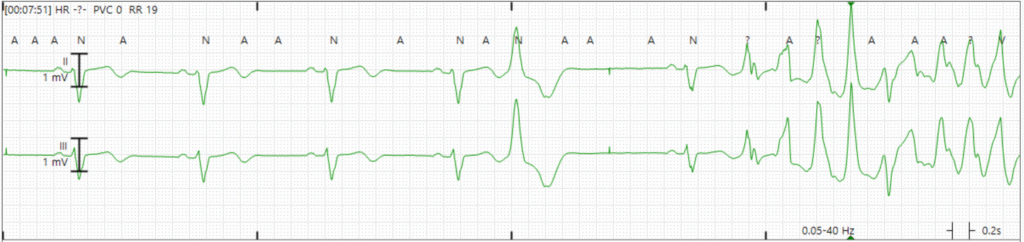
The patient had R on T phenomenon resulting in TdP which rapidly degenerated into ventricular fibrillation (soon after this rhythm strip). Since she was being monitored on cardiac telemetry, the nursing team activated code blue and began ACLS. The patient was quickly intubated and defibrillated, and brought to the ICU. She had no recurrent arrhythmias the remainder of the night, and dofetilide was obviously discontinued.
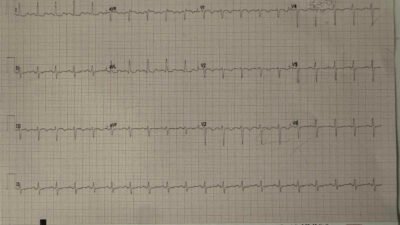
In the middle of the next day ( = hospital day 2), while still intubated and sedated — she had resting sinus bradycardia in the low 40s which was initially uneventful. You can see on this compressed strip showing 12 minutes of telemetry that she had not been having any PVCs.
Figure 5: Sinus bradycardia on telemetry

However, she abruptly began having PVCs and runs of self-terminating TdP.
Figure 6: PVCs and recurrent TdP
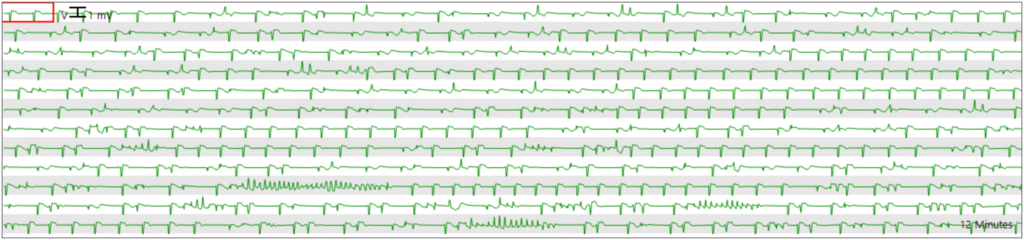
Figure 7: Zoomed in view of a short run of TdP
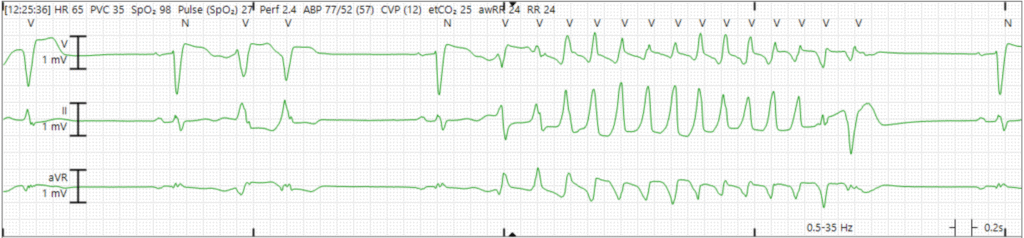
The clinical team rapidly gave the patient 4 g of intravenous magnesium, but the PVCs and TdP persisted.
Take a moment and ask yourself, what would you do next?
Answer: Increase the heart rate. Overdrive pacing is a highly effective way to suppress TdP. The exact mechanism of benefit is not entirely clear, but is probably due to the dual impact of tachycardia to: 1) Shorten the QT, and 2) Reduce repolarization dispersion (that is, the regional heterogeneity in states of repolarization across different regions of the heart at a given time in the cardiac cycle).
How can you increase the heart rate? Two commonly employed techniques are 1. temporary transvenous pacemaker or 2. continuous infusion of a beta-1 agonist. Formerly, the most commonly used beta-1 agonist was isoproterenol, but now it is so expensive that dobutamine is more common. However, both of these methods will generally take 20 minutes or more to initiate. Twenty minutes is a long time when you’re standing bedside watching recurrent salvos of TdP on the monitor.
What can we do for the patient right now using the tools we have in the crash cart? Atropine! Atropine is always in the crash cart, and it can raise the heart rate immediately while you wait for pharmacy to prepare isoproterenol. This is not widely taught as a treatment for TdP, but it is often extremely effective. There is nothing magic about isoproterenol, it just happens to be a titratable IV medication that can be used to sustain an elevated heart rate relatively easily.
Back to the case:
The patient received atropine 1 mg IV from the crash cart and look what happened:
Figure 8: Telemetry before and after atropine
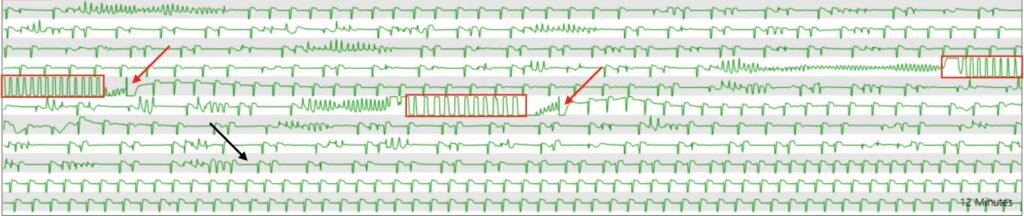
Looking through this segment which represents 11 minutes (I cut the top line off to focus on the important parts), we see recurrent salvos of TdP. The red rectangles highlight chest compression artifact during episodes of sustained TdP. The red arrow heads point to artifact from external defibrillation. The black arrow head points to the time that atropine was given.
The patient was subsequently started on isoproterenol which was titrated to achieve target heart rate of 80 beats per minute. This was titrated off over 48 hours as the dofetilide washed out. The patient did not have any recurrent TdP.
Learning Points:
- In addition to IV magnesium, think about atropine when treating Torsades de Pointes if there is associated bradycardia.
- QTc is harder to interpret in patients with wide QRS.
Polymorphic VT with prolonged QT (Torsades de Pointes)
•Usually self terminating, may progress to v fib
•“Pause Dependent” (worse with bradycardia)
•Treatment: correct electrolytes (K, Mg)
•At risk of torsade: Mg, 2g over 15 min
•Active VT:
–Cardiovert
–Mg, 2g over 30-60 sec, max 6g
•get Serum K to 4.0 to 4.5
•Lidocaine may prevent PVCs, won’t prolong QT like others
•Overdrive pacing (100-140): Lowest pacing rate that prevents PVC’s
•Isoproterenol (beta 1 and 2 agonist!), if pacing cannot be started immediately.
•Dobutamine is a substitute and is cheaper now.
•Prevents pause-dependent LQTS torsade by increasing HR
•Use if Torsades appears to be pause dependent
–If it is pause dependent, then it is almost always acquired Long QT (due to drugs or electrolytes)
–Underlying bradycardia prior to tachycardia (“Pause dependent”)
•Beware Catecholaminergic Polymorphic VT
–This type of polymorphic VT is worsened with the use of a beta-1 agonist!)
–It is also worse with cardioversion (due to catecholamine surge)
–Shock and beta blockade together
–ECG without long QT
= = =
Ibutilide use to facilitate cardioversion of atrial fib in the ED
Ibutilide can be used to facilitate cardioversion in patients in whom electricity alone is not effective. See this article (not an ED use), described in detail below: Facilitating Transthoracic Cardioversion of Atrial Fibrillation with Ibutilide Pretreatment
And this is a description of the use of ibutilide in the ED (they were much less restrictive than the original study, described below, with few complications): Ibutilide Effectiveness and Safety in the Cardioversion of Atrial Fibrillation and Flutter in the Community Emergency Department
Facilitating Transthoracic Cardioversion of Atrial Fibrillation with Ibutilide Pretreatment. Oral H et al., New Engl J Med June 17, 1999; 340(24):1849-54.
Abstract
Background. Atrial fibrillation cannot always be converted to sinus rhythm by transthoracic electrical cardioversion. We examined the effect of ibutilide, a class III antiarrhythmic agent, on the energy requirement for atrial defibrillation and assessed the value of this agent in facilitating cardioversion in patients with atrial fibrillation that is resistant to conventional transthoracic cardioversion.
Methods. One hundred patients who had had atrial fibrillation for a mean (±SD) of 117±201 days were randomly assigned to undergo transthoracic cardioversion with or without pretreatment with 1 mg of ibutilide. We designed a step-up protocol in which shocks at 50, 100, 200, 300, and 360 J were used for transthoracic cardioversion. If transthoracic cardioversion was unsuccessful in a patient who had nreceived ibutilide pretreatment, ibutilide was administered and transthoracic cardioversion attempted again.
Results. Conversion to sinus rhythm occurred in 36 of 50 patients who had not received ibutilide (72 percent) and in all 50 patients who had received ibutilide (100 percent, P<0.001). In all 14 patients in whom transthoracic cardioversion alone failed, sinus rhythm was restored when cardioversion was attempted again after the administration of ibutilide. Pretreatment with ibutilide was associated with a reduction in the mean energy required for defibrillation (166±80 J, as compared with 228±93 J without pretreatment; P<0.001). Sustained polymorphic ventricular tachycardia occurred in 2 of the 64 patients who received ibutilide (3 percent), both of whom had an ejection fraction of 0.20 or less. The rates of freedom from atrial fibrillation after six months of follow-up were similar in the two randomized groups.
Conclusions. The efficacy of transthoracic cardioversion for converting atrial fibrillation to sinus rhythm was enhanced by pretreatment with ibutilide. However, use of this drug should be avoided in patients with very low ejection fractions. (N Engl J Med 1999;340:1849-54.) *
Important comments:
The dose was 1 mg over 10 minutes IV.
They included patients who had had a fib for less than 48 hours
They excluded patients with a fib for longer than 48 hours unless they proved, by TE echo, to not have an atrial clot OR unless they anticoagulated them for 3 weeks first
Therefore, our patients who have been in afib < 48 hours, or who have been on anticoagulants, apply
They excluded anyone with a QTc > 480ms because ibutilide can lead to torsade
The dose was 1mg over 10 minutes
In the 2 patients who went into torsades (both with low ejection fractions, <20%), both were easily controlled.
The authors recommend not using ibutilide for this indication in stable patients if the EF is < 30%. However, it is still an option in unstable patients.
Ibutilide increased the QTc significantly (432+/-37 before vs. 482+/-49 afterward)
Although the point is not discussed in the paper, I would not send such a patient home unless the QT is corrected.
Finally, I always first use electrical cardioversion without ibutilide. As in this study, I only use ibutilide if electricity does not work.
======================================
MY Comment, by KEN GRAUER, MD (2/25/2026):
Assessing the potential risk of TdP (Torsades de Pointes) is always challenging — but it is especially so when there is QRS widening and bradycardia (with both of these findings present in today’s case).
- Bradycardia results in longer R-R intervals. Since the R-R interval before a given QRS complex influences how long the QT interval of that QRS complex will be — slower heart rates result in an increased duration of the QT interval for subsequent beats (and therefore a lengthening of the subsequent “vulnerable” period). It is for this reason that when we are concerned about the “risk” of TdP — that it is probably not a good idea to “correct” for the QT interval with heart rates below 60/minute (since slow heart rates automatically increase the risk of Torsades).
- I’d therefore add to Dr. Frick’s Learning Points the folllowing: i) Increasing degrees of bradycardia may substantially increase the risk of Torsades; — and, ii) Correction formulas for QTc estimation become less accurate with slow heart rates (such that less, if any correction of the measured QT might be the best approach for assessing the “QTc” when the heart rate is less than 60/minute).
= = =
How to estimate the QTc when the QRS is wide?
- Today’s case by Dr. Frick highlights the clinical importance of QTc estimation (relating in this case to Dofetilide dosing).
- We’ve added a quick link to our QTc calculator — in the Menu at the top of every page in Dr. Smith’s ECG Blog (Figure-1). This calculator facilitates QTc estimation using the most widely accepted formulas for both normal and prolonged QRS complexes (Simply slide the calculator over to the left to access the application when the QRS is wide).
= = =
Figure-1: Quick link to the QTc Calculator in the Menu at the TOP of every page in Dr. Smith’s ECG Blog.

= = =
=