Sent by anonymous, written by Pendell Meyers
A middle aged man presented with acute shortness of breath. Apparently he denied chest pain. Other than tachycardia, Other than slight tachycardia, vitals were within normal limits (including oxygen saturation).
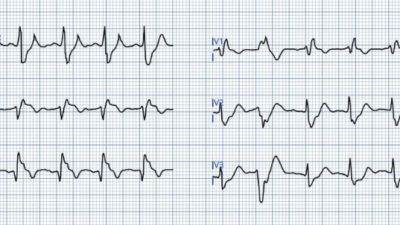
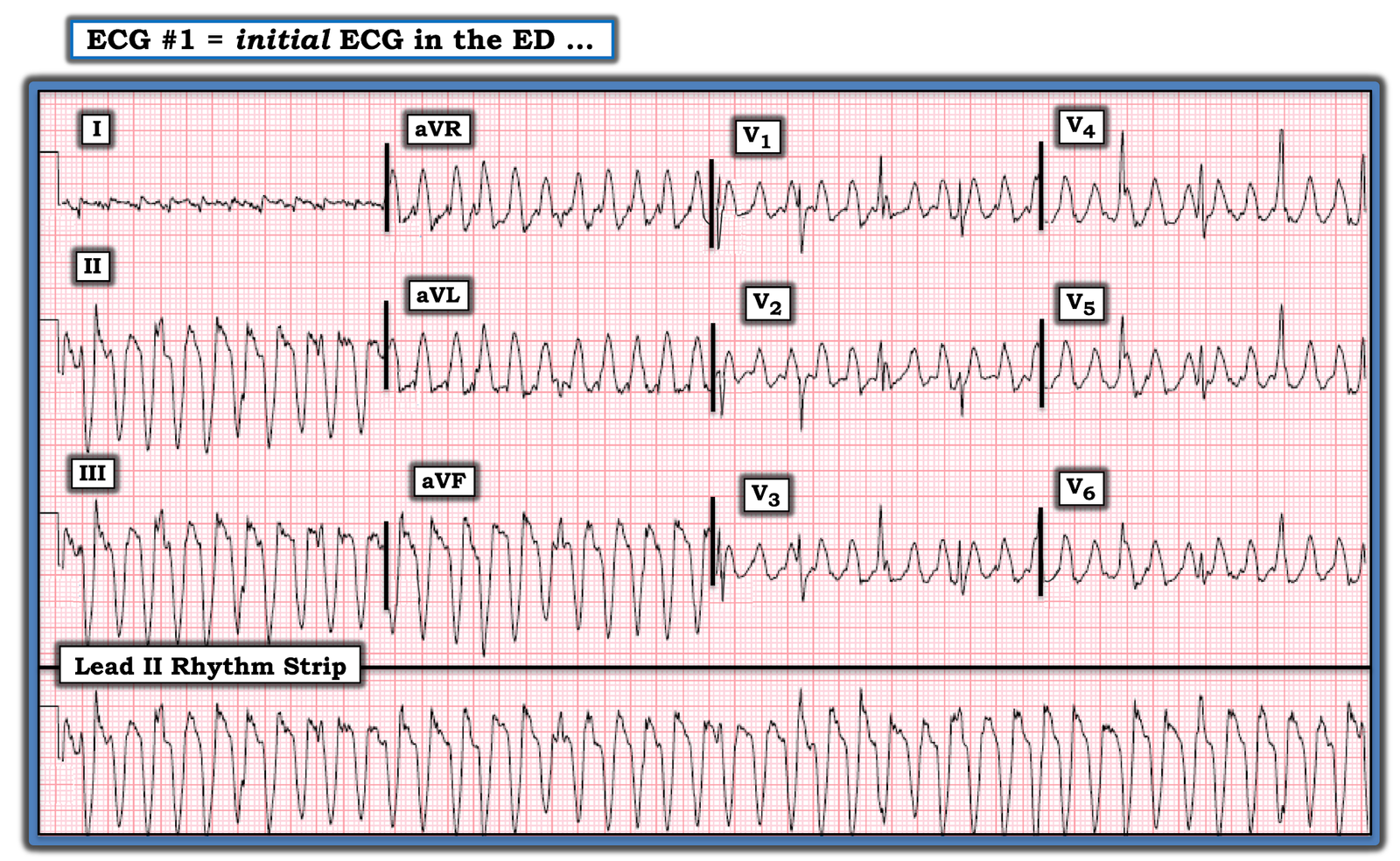
Here is his first ED ECG:
 |
| What do you see? |
Findings:
– Sinus tachycardia
– Poor R wave progression
– STD in leads V3 and V4
– there is almost an appearance of STE in V6, but it is not definite
– there is also slight STE in aVL with slight reciprocal STD in inferior leads
Impression: Diagnostic of posterior OMI [and the subtle lateral involvement (aVL) supports this] until proven otherwise. STD maximal in V1-V4 (in this case V3-V4) is in my opinion the single best way to identify posterior OMI on the anterior 12 lead ECG. As always, takotsubo cardiomyopathy and focal pericarditis can mimic OMI, but takotsubo almost never mimics posterior MI, and both are diagnoses of exclusion after a negative cath.
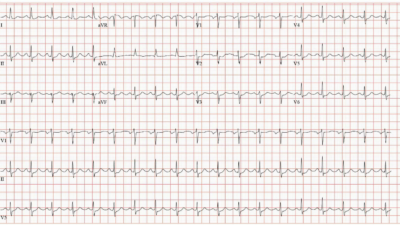
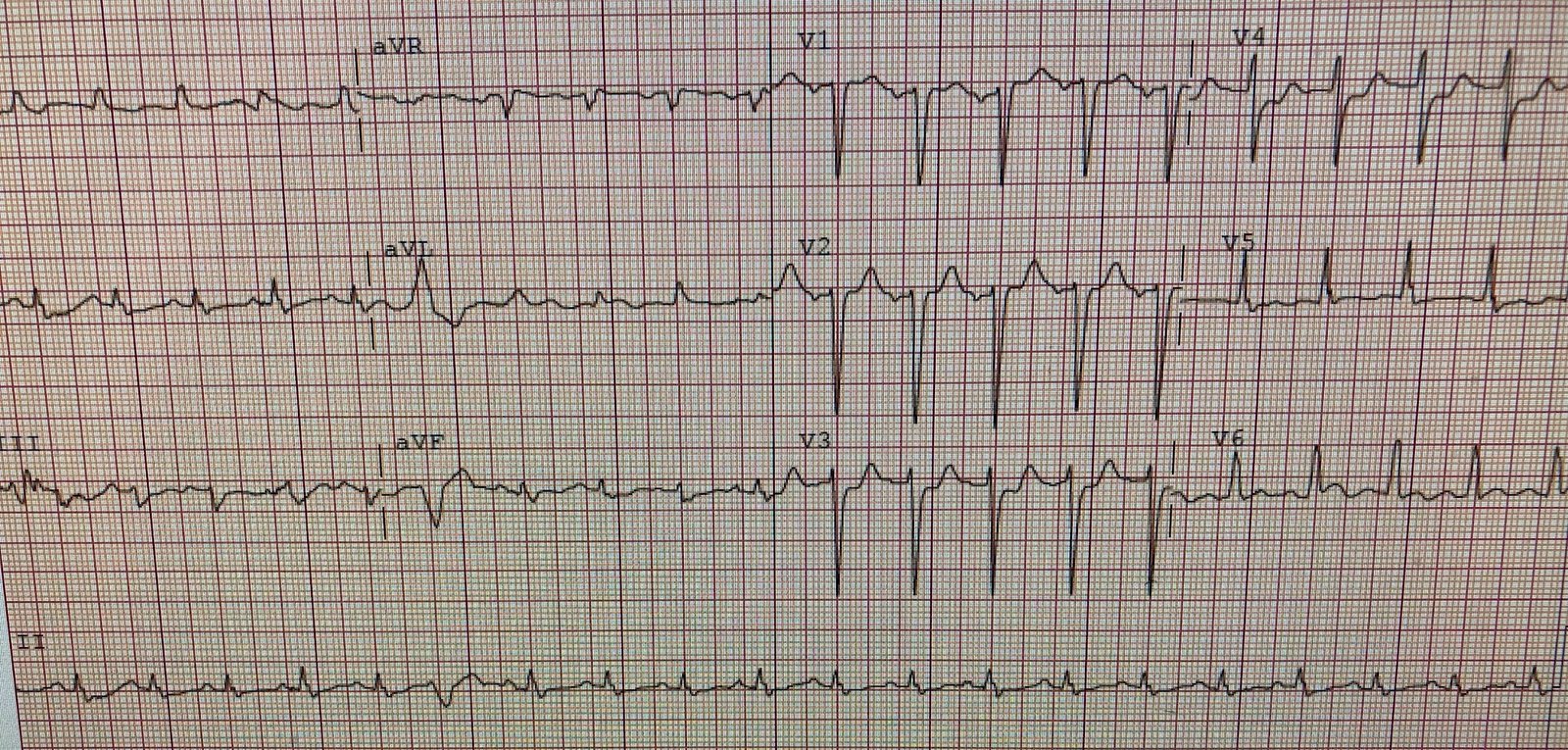
His first troponin returned “elevated” (level not available). Another ECG was recorded based on the elevated troponin and ongoing symptoms:
 |
| The heart rate is slightly lower. The posterior and lateral OMI findings persist, with STD maximal in V3 and V4. |
The provider contacted cardiology to discuss the case, but cardiology “didn’t think it was a STEMI, didn’t think he needed emergent cath.”
He was admitted to the cardiology floor and diagnosed with an NSTEMI. About two hours after admission, he suffered a cardiac arrest (whether it was VF/VT or PEA is not available) and expired.
Like other cases on this blog who died before cath was done, I cannot prove that this patient had OMI. But I would submit to you that it is overwhelmingly likely that this was the case. Could this outcome have been prevented with emergent cath? I think there is a significant chance that it could have been.
Learning Points:
STD maximal in V1-V4 (in the absence of another reason for STD like appropriate discordance from an abnormal QRS complex) is concerning for posterior OMI until proven otherwise.
When the anterior 12 lead is not convincing for posterior OMI, posterior leads may help. When the anterior 12 lead is convincing as in this case, posterior leads don’t add much and may actually mislead you or the cardiologist.
Canto et al. (JAMA 2000) showed that 1/3 of patients with STEMI, and 1/3 of patients with NSTEMI, present without chest pain. https://jamanetwork.com/journals/jama/article-abstract/192825
Further discussion:
Dr. Smith and I will soon be publishing a paper showing that expert ECG interpretation is greatly superior to standard STEMI criteria. “ST depression maximal in V1-V4” is one of the key features we found that helped experts identify OMI that the STEMI criteria missed. We submitted this study first to JACC, who rejected it because “No one is strictly limited by STEMI criteria. The 4th universal definition mentions ST depression, posterior MI, and T wave changes.”
We also know for a fact that STEMI criteria misses about half of OMIs, at least 25% of “NSTEMIs” are found to have an occluded infarct artery at next day angiography (and since many occluded arteries open over the subsequent 24 hours, this underestimates the number of OMI), and LCX occlusions are the most commonly missed (OMIs that usually cause posterior and/or lateral ECG findings). See OMI Manifesto for references.
Despite the fact that there is no publication that has candidly recorded how cardiologists actually interpret the STEMI guidelines, we all know our own experience at our institutions as to how the majority of cardiologists interpret them in actual practice. Furthermore, we receive cases from around the world that give us additional evidence that this problem is likely highly prevalent. The whole paradigm is literally called “STEMI” vs. “NSTEMI.” It’s not surprising, and it’s arguably not even their fault – for the majority of physicians who manage patients with symptoms of ACS, the cath lab might as well be called the “STEMI criteria lab,” because it seems to us and our audience that in most hospitals around the world, only a patient with an ECG with full blown obvious STEMI criteria (which does NOT include anything about the posterior wall) is likely to be going there emergently.
At the same time that the editorial board of JACC rejects our paper because they claim that all cardiologists are very much on board with taking NSTEMI patients with possible posterior MI or ST depression to the cath lab, we are left hanging in reality where it is very difficult to get posterior MI recognized by cardiology as an entity.
We have countless cases of missed posterior MI on this blog, including these:
Interventionalist at the Receiving Hospital: “No STEMI, no cath. I do not accept the transfer.”
“It isn’t a STEMI,” so cath lab refusal (again). Were they right?
A middle aged man with ST depression and a narrow window of opportunity
A man in his 50s with 2 hours of chest pressure
A woman in her 70s with bradycardia and hypotension
You have two hours to save this patient’s life
A man in his early sixties with palpitations
A female in her 60s with sudden chest pressure
How much time are you willing to wait for OMI to become STEMI (if it ever does)?
Chest pain and Concordant ST Depression in a patient with aortic valve and previously normal angiogram
Right Bundle Branch Block and ST Depression in V1-V3. Is that normal? And a complication.
===================================
MY Comment by KEN GRAUER, MD (10/11/2020):
===================================
It cannot be stated any clearer than what Dr. Meyers states above. I will paraphrase ==> “the STEMI criteria that all-too-many cardiologists rigidly adhere to, will miss as many as half of all OMIs — and — at least 25% of NSTEMIs are later found to have been missed OMI”.
- Our blog features countless cases of missed OMI — with (as per Dr. Meyers) — missed posterior MI from acute LCx occlusion being the site most commonly overlooked.
Today’s post features yet one more case of a missed posterior OMI — with tragic consequence because the cardiology team “didn’t think this was a STEMI” — and “didn’t think the patient needed emergent cath”.
- Our GOALS for continuing to post these cases in Dr. Smith’s ECG Blog are: i) To support readers in their quest to continually enhance their ECG interpretation skills (many of these readers already having attained expertise in recognizing OMIs); and, ii) In our hope that some of these missed cases will get back to those cardiologists who continue to ignore the possibility that an ECG not satisfying millimeter STEMI criteria might nevertheless be diagnostic of OMI ( = acute coronary Occlusion-based MI) — and, in need of prompt cath and reperfusion.
PEARL #1: The patient in today’s case presented with acute shortness of breath but no chest pain. The lack of chest pain in the history could have been part of the reason the cardiology team did not activate the cath lab. That said, it is important to realize that — Not all patients with acute MI have chest pain! I’ve previously emphasized this important truism (in My Comment at the bottom of the page in the September 15, 2020 post in Dr. Smith’s ECG Blog) — but for clarity, I’ve consolidated key findings from the Framingham Studies regarding the incidence of Silent MI in Figure-1.
- As highlighted in the 3rd bullet in Figure-1 — shortness of breath is the most common non-chest-pain presenting symptom of acute MI. The absence of chest pain in today’s patient should not have dissuaded the cardiology team from at least considering OMI, given how suggestive ECG findings were for that diagnosis.
Figure-1: Key points from the Framingham Studies regarding the entity of “Silent” MI (See text).
Dr. Meyers highlights above the ECG findings in today’s case that should suggest acute posterior OMI until proven otherwise. I focus my comments on some additional points for facilitating ECG recognition.
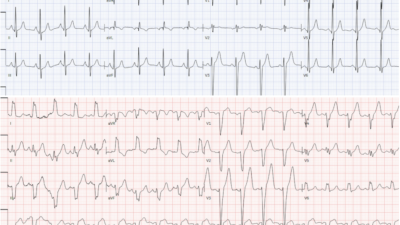
- For clarity — I’ve put both tracings in today’s case together in Figure-2.
Figure-2: The 2 ECGs in today’s case (See text).
MY Thoughts on ECG #1: Admittedly, the quality of resolution for the 2 tracings in Figure-2 is suboptimal — with variation of QRST morphology from one-beat-to-the-next in many leads making interpretation a challenge. That said — the diagnosis should still be made.
- The rhythm in ECG #1 is sinus tachycardia at 115-120/minute. A PVC is seen (in leads aVR,aVL,aVF). The PR interval is normal. QRS duration is not prolonged. The QTc may be upper normal, or slightly prolonged. There is low voltage in the limb leads. The frontal plane axis is normal (about +20 degrees). There is borderline voltage for LVH by Peguero Criteria.
Regarding Q–R–S–T Changes in ECG #1:
- There are no Q waves.
- R wave progression is slightly delayed (ie, Transition occurs between leads V4-to-V5). That said — a small-but-define initial r wave is seen in leads V1, V2 and V3.
- ST-T wave abnormalities are present. There is nonspecific ST-T wave flattening in several limb leads (though I fully acknowledge the deceptive variation in ST-T wave morphology in these limb leads). That said — what is indisputable is that maximal ST depression is consistently seen in all complexes in leads V3 and V4!
- As per Dr. Meyers — there is also suggestion of slight ST elevation in lead V6 (and possibly also in lead aVL) — albeit suboptimal resolution with variation in ST-T wave morphology makes validity of this finding uncertain.
PEARL #2: While fully acknowledging the above noted beat-to-beat variation in ST-T wave morphology — each of the QRS complexes in leads V3 and V4 are consistent in showing that ST depression is maximal in these chest leads. It is this consistency that tells us this is a real finding!
PEARL #3: I’ve described the Mirror Test on a number of occasions in Dr. Smith’s ECG Blog (Please see My Comment in the September 21, 2020 post). This simple visual aid facilitates recognition of acute posterior MI. By flipping the ECG over and holding it up to the light — you see the mirror-image of the anterior leads. Since the LV posterior wall is oppositely directed to the anterior wall — this mirror-image indirectly assesses the posterior wall.
- DOESN’T the mirror-image of leads V3 and V4 in Figure-2 show an ST-T wave shape that looks like an acute MI? (ie, with downward coving ST elevation that ends with T wave inversion).
PEARL #4: Remember the principle of “neighboring leads”. Given certainty that the maximal ST depression in leads V3 and V4 is real — we can often pick up additional leads with less striking abnormalities that we nevertheless know are real because they are “neighboring” leads.
- Note that all 4 of the complexes ST in lead V2 show ST straightening, with marked angulation as the ST segment transitions to the ascending limb of the T wave (thin RED lines in lead V2). While I admittedly would not have known what to do with this subtle finding IF it was an isolated abnormality — given its proximity to leads V3 and V4 (that we know are abnormal) — there is little doubt that this subtle ST straightening and abrupt angulation in lead V2 also is abnormal.
MY Thoughts on ECG #2: The 2nd ECG in this case ( = ECG #2) was obtained in the ED some time after ECG #1 (although we are not told how long this was after ECG #1, nor whether there was a change in the patients symptoms or condition).
- There is a definite change between these 2 tracings — although it is EASY to overlook unless one puts both tracings together (as is done in Figure-2) — and compares tracings by going lead-by-lead from one tracing to the next.
- There is still sinus tachycardia in ECG #2 — although the heart rate has now slowed to ~100/minute.
- There are no PVCs in ECG #2.
- The subtle suggestion of slight ST elevation in leads aVL and V6 is no longer seen.
- The main difference between these 2 tracings — is that the upright T waves in leads V1-thru-V4 in ECG #1 are practically gone! (except for a tiny, residual upright T wave in lead V2 of ECG #2). Depending on clinical circumstances — this change in T wave morphology in no less than 4 leads could either be rate-related (ie, the slower heart rate in ECG #2) — or could possibly reflect a dynamic ST-T wave change in this patient with ongoing OMI.
BOTTOM Line: In view of the 2 serial tracings in today’s case — one has to at least consider a posterior OMI in this patient with new dyspnea until proven otherwise.